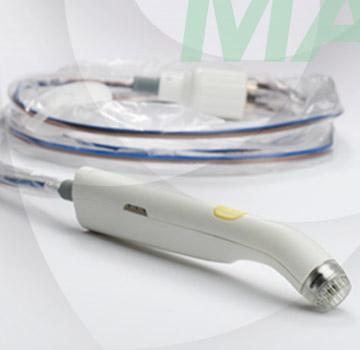
Image courtesy of Dune Medical Devices
January 9, 2015 — Dune Medical Devices announced U.S. Food and Drug Administration (FDA) approval of an updated version of its flagship product, MarginProbe, a medical device that enables real-time detection of cancer at the surface of excised tissue specimens during breast-conserving cancer surgery.
Surgeon feedback, innovative design ideas and superior miniaturization engineering were the driving forces behind the development of MarginProbe 1.2. This new version, which utilizes the same FDA-approved diagnostic technology as version 1.1, is focused on providing superior functionality, portability and overall ease of use.
Feature enhancements for MarginProbe 1.2 include:
- Reduced size & weight: The new unit is in tabletop form (29 x 43 x 38 cm in size) and weighs 35 lbs. making it 75-percent lighter and much easier to handle.
- Brighter screen: The screen’s brightness has been enhanced to provide surgeons a clearer screen image.
- Wider viewing angle: With MarginProbe 1.2, surgeons will have the ability to clearly view the screen from anywhere in the operating room without encountering a glare, pixelation or a blind spot.
- Improved on-screen notices: Ensuring key alerts are easier to notice in the OR ambience.
- Startup time cut in half: The startup time for MarginProbe 1.2 is reduced from approximately three minutes to 90 seconds.
- Improved service time: Due to its new reduced size, technicians are able to provide faster service, reducing down time in case service is needed.
For more information: www.dunemedical.com


 May 22, 2026
May 22, 2026 









