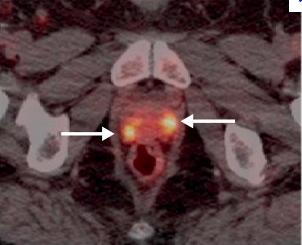
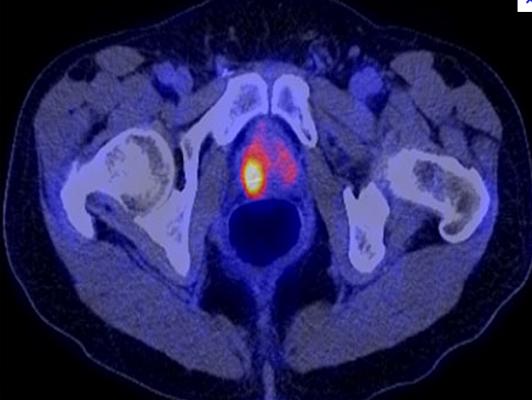
September 14, 2012 — The U.S. Food and Drug Administration (FDA) has approved the production and use of Choline C-11 injection, a positron emission tomography (PET) imaging agent used to help detect recurrent prostate cancer. Choline C-11 injection is administered intravenously to produce an image that helps to locate specific body sites for follow-up tissue sampling and testing in men with recurrent prostate cancer.
PET imaging with Choline C-11 injection is performed in patients whose blood prostate specific antigen (PSA) levels are increasing after earlier treatment for prostate cancer. An elevated PSA result suggests that prostate cancer may have returned, even though conventional imaging tests, such as computed tomography (CT), have not shown any signs of cancer. PET imaging is not a replacement for tissue sampling and testing.
Choline C-11 injection must be produced in a specialized facility and administered to patients shortly after its production. While PET imaging with Choline C-11 injection has been performed at a few facilities over the past several years, none of these facilities were approved by the FDA to manufacture the agent. The Food and Drug Administration Modernization Act directed the agency to establish appropriate approval procedures and current good manufacturing practice requirements for all PET products marketed and used in the United States. The Mayo Clinic is now the first FDA-approved facility to produce Choline C-11 injection.
“Choline C-11 injection provides an important imaging method to help detect the location of prostate cancer in patients whose blood tests suggest recurrent cancer when other imaging tests are negative,” said Charles Ganley, M.D., director of the Office of Drug Evaluation IV in FDA’s Center for Drug Evaluation and Research. “The FDA’s approval of Choline C-11 injection at the Mayo Clinic provides assurance to patients and healthcare professionals they are using a product that is safe, effective and produced according to current good manufacturing practices.”
The safety and effectiveness of Choline C-11 injection were verified by a systematic review of published study reports. Four independent studies examined a total of 98 patients with elevated blood PSA levels but no sign of recurrent prostate cancer on conventional imaging. After PET imaging with Choline C-11, the patients underwent tissue sampling of the abnormalities detected on the PET scans.
In each of the four studies, at least half the patients who had abnormalities detected on PET scans also had recurrent prostate cancer confirmed by tissue sampling of the abnormal areas. PET scan errors also were reported. Depending on the study, falsely positive PET scans were observed in 15 percent to 47 percent of the patients. These findings underscore the need for confirmatory tissue sampling of abnormalities detected with Choline C-11 injection PET scans. Aside from an uncommon, mild skin reaction at the injection site, no side effects to Choline C-11 injection were reported.
Choline C-11 injection is manufactured and distributed by the Mayo Clinic PET Radiochemistry Facility in Rochester, Minn.
For more information: www.fda.gov/Drugs/DevelopmentApprovalProcess/Manufacturing/ucm085783.htm


 May 29, 2026
May 29, 2026 









