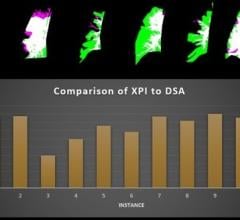
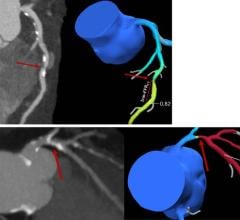
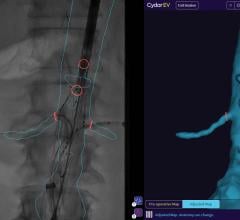
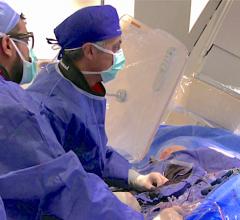
The GE Healthcare Discovery IGS 730 angiography imaging system.
March 26, 2012 – GE Healthcare unveiled two recently U.S. Food and Drug Administration (FDA)-cleared angiography systems offering mobility and advanced imaging for interventional cardiology at the American College of Cardiology's (ACC) 61st Annual Scientific Session in Chicago this week.
Specifically, GE highlighted its recently FDA-cleared Discovery IGS 730 system, the first interventional X-ray system designed to capture the advantages of both floor- and ceiling-mounted systems, and the mobility of a mobile C-arm. Initially introduced last fall during RSNA 2011, the Discovery system was designed to address the trade-off departments have historically needed to make between ceiling- and floor-mounted systems, eliminating the need to compromise on patient access and image quality. This laser-guided, motorized mobile gantry allows complete access to the patient and unlimited parking capability, while creating sterility for a flexible operating room (OR) environment. The unique gantry comes with a new wide bore design, which allows for steep angles, ease in 3-D acquisition, especially for large patients.
GE will also introduce the FDA-approved Innova IGS 520/530 with the OR option to expand the use of its Innova imaging system in the hybrid OR setting. With specifically designed table and accessories, the Innova IGS 520/530 OR enables both transcatheter and open surgical procedures in one room.
GE also highlighted its Innova HeartVision solution to help support planning and guidance of structural heart procedures, especially trans-aortic valve replacement (TAVR) procedures. The HeartVision function fuses real time 2-D X-ray images and 3-D models, such as the 3-D cardiac model, from multiple modalities. It can be synchronized with electrocardiogram (ECG) gating and can help compensate for patient heart and respiratory motion throughout the procedure for quality treatment. Innova HeartVision can reduce procedure time and dose, and increase physician confidence by providing the visual tools to make challenging interventions more accessible.
“Interventional cardiology procedures are becoming increasingly complex, and now may be performed in a variety of settings in the hospital, including the operating room. GE Healthcare is revolutionizing cardiac imaging with solutions that adapt to a wider array of environments,” said Reaz Rasul, general manager, interventional cardiology, GE Healthcare - detection and guidance solutions. “With the development of Discovery IGS 730 and Innova IGS 520/530 OR, we are arming physicians with solutions that combine cutting-edge imaging with greater versatility to meet more clinical needs.”
To see a video of the system in operation when it was first unveiled at RSNA 2011, click here.
For more information: www.gehealthcare.com


 March 05, 2026
March 05, 2026