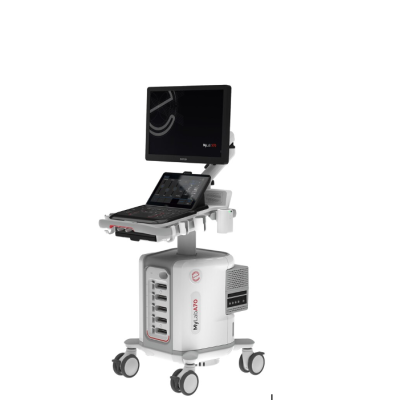
The Esaote MyLab A70 ultrasound system
Aug. 22, 2025 — Esaote recently announced that the MyLab A50 and MyLab A70 ultrasound systems have received FDA approval. Previously unveiled to the international medical community, these advanced platforms are now cleared for distribution in the United States, underscoring Esaote's ongoing commitment to innovation in diagnostic imaging.
MyLab A50 and MyLab A70 ultrasound systems are engineered to deliver exceptional flexibility and performance across a wide range of clinical environments. These new models are designed to be highly portable, featuring compact sizes and battery operation to meet the mobility needs of healthcare practitioners.
According to Thomas Will, Director of Ultrasound Sales at Esaote North America "the new A-series emphasizes user experience with a diverse range of interface options, including both a conventional and touch control panel. The devices boast an intuitive, easy-to-clean design that allows clinicians to operate efficiently and confidently."
The MyLab A50 and MyLab A70 are equipped to handle an array of clinical applications, from routine diagnostics to advanced imaging techniques. They support the latest ultrasound functionalities, such as liver elastography attenuation imaging, along with comprehensive cardiology tools like strain analysis, enabling detailed and multiparametric assessments.
Harnessing artificial intelligence and cutting-edge imaging advancements, these systems empower healthcare providers to make precise, informed diagnoses with greater confidence.
Marking a significant milestone for Esaote's renewed brand mission, the introduction of the MyLab A50 and MyLab A70 underscores the company's dedication to improving patient outcomes through innovative technology and compassionate care.


 May 12, 2026
May 12, 2026 









