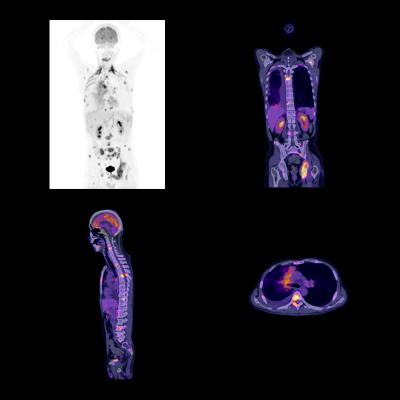
September 13, 2017 — Blue Earth Diagnostics announced the upcoming oral presentation of initial results from the FALCON clinical trial of fluciclovine (18F) positron emission tomography/computed tomography (PET/CT) at the 2017 American Society for Radiation Oncology (ASTRO) annual meeting Sept. 24-27 in San Diego. The FALCON trial evaluated the impact of F-18 PET/CT on patient management in biochemically recurrent prostate cancer patients scheduled for salvage radiation therapy treatment with curative intent. Trial results will be presented by Eugene Teoh, M.D., Oxford University Hospitals NHS Trust, Oxford, U.K.
Blue Earth Diagnostics is also hosting an Industry-Expert Theater event, “A Novel Option for Biochemically Recurrent Prostate Cancer Localization,” with invited speaker Abhishek Solanki, M.D., assistant professor of radiation oncology, Loyola University School of Medicine, Chicago, which will be held on Tuesday, Sept. 26.
For more information: www.blueearthdiagnostics.com


 May 27, 2026
May 27, 2026 









