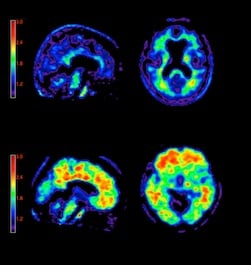
Several positron emission tomography (PET) imaging agents are being developed to visualize Alzheimer’s disease in the living brain to accurately diagnose patients and develop effective treatments. The only way to confirm the presence of the disease currently is by examining post-mortem samples of the patient’s brain. Experts predict these agents may soon receive U.S. Food and Drug Administration (FDA) clearance.
It is estimated 18 million people worldwide have Alzheimer’s disease, and that number could double by 2025. There is currently no definitive early detection method for the disease. Symptoms of Alzheimer’s usually do not appear until after the disease is well established and by that time, damage has already been caused to the brain.
Post-mortem samples of the patient’s brain tissue are used to positively identify the disease by the presence of beta-amyloid plaques – a root cause of Alzheimer’s. These plaques group together and kill neurons. However, the inability to image the soft tissue disease inside a living brain has hampered the ability of researchers to track its progress in living patients or monitor the effect of potential treatments.
PET May Offer the Key
Researchers are now advancing PET as an effective method of early detection. Three studies presented in June at the Society of Nuclear Medicine (SNM) annual meeting provide new insights into the development of Alzheimer’s disease and open the door to future clinical screening and development of treatments.
“Molecular imaging of Alzheimer’s disease might be available in the next year,” predicted Michael Graham, M.D., Ph.D., during a press conference on key molecular imaging research at the SNM meeting. “There is fairly good evidence you can image early stages of Alzheimer’s before the disease progresses. It opens up the possibility of new therapies for treatment. This has been a huge limitation.”
He said several agents are being developed. The most promising include Pittsburgh compound B (PiB), developed by the University of Pittsburgh, and AV45, owned by Eli Lilly and Company.
These imaging agents are key in developing treatments for Alzheimer’s, said Chet Mathis, Ph.D., professor of radiology, department of pharmacology and chemical biology, University of Pittsburgh, during a presentation at SNM. He helped develop PiB, which he explained is a carbon 11 compound that uses neutrally charged thioflavin T, an established histological stain used to detect beta-amyloid.
Clinical studies of PiB have successfully identified patients with probable Alzheimer’s from healthy patients and predicted the likelihood of disease progression in those with mild cognitive impairment. However, widespread use of the 11-C compound is limited, due to its short 20-minute half-life and the need for an onsite cyclotron.
Development of 18-F Agents
One of the major questions in Alzheimer’s imaging has been which imaging agent is best for amyloid plaque screening. Due to the limitations of PiB, 18-F amyloid imaging agents are seen as the most likely to move forward into clinical practice in the near future. Two of the current studies are gauging the benefit of using 18-F labeled tracers (18-F florbetaben and 18-F florbetapir), which are proving to be good predictors of progression to Alzheimer’s disease.
Lilly is developing the imaging agent florbetapir 18-F AV-45 (trade name is Amyvid). Hank Kung, Ph.D., professor of radiology and pharmacology, department of radiology, University of Pennsylvania, helped develop the compound and spoke about it and other compounds at the SNM meeting.
Both Kung and Mathis said developing agents that can pass through the blood-brain barrier is difficult. In addition, the agent needs to have an affinity to bind with amyloid plaque. The third key component is the need to have a good signal-to-noise characteristic, where the agent is still clearly visible on images and remains bound to the plaques after it has washed out of other parts of the brain not affected by plaques. Mathis said this is one of the most difficult features to find when developing these agents.
Kung screened more than 1,000 compounds over seven years before finding AV-45. Avid Radiopharmaceuticals successfully completed a Phase III FDA clinical trial for the agent in 2010. Eli Lilly and Company purchased the company in November 2010.
In January 2011, an FDA advisory committee unanimously recommended that Avid’s AV-45 PET agent be approved for use, but required the company to develop clear guidelines establishing when a test has identified enough amyloid plaque to make a positive diagnosis of Alzheimer’s. Lilly and Avid Radiopharmaceuticals received a complete response letter from the FDA in March 2011, which focused on the need to establish a reader training program for market implementation to ensure reader accuracy and consistency of interpretations of existing AV-45 scans. The company expects submission for final FDA review of AV-45 in the second half of 2011.
GE Healthcare is developing a flutemetamol 18-F agent that is currently in Phase III development. Three clinical abstracts examining the efficacy of flutemetamol amyloid imaging in the early detection of Alzheimer’s were featured in July at the 2011 Alzheimer’s Association International Conference on Alzheimer’s Disease (ICAD) in Paris. A recently published study[1] showed complete agreement between visual reads of 18-F flutemetamol PET scans and histology.
Bayer is developing 18-F florbetaben, which is in a Phase III trial of 400 patients with and without dementia. The trial is expected to be completed in 2011, but data is not expected to be finalized until 2014. Data so far show a sensitivity of 80 percent and a specificity of 91 percent in identifying patients with Alzheimer’s from healthy controls.[2]
AstraZeneca is working on an agent called AZD4694, a benzofuran-derived radioligand containing fluorine 18. The agent is currently in a Phase II trial, which the company expects to complete in 2011.[3]
References:
1. David A. Wolk, et al. “Association Between In Vivo Fluorine 18–Labeled Flutemetamol Amyloid Positron Emission Tomography Imaging and In Vivo Cerebral Cortical Histopathology.” Archives of Neurology. Published online July 11, 2011. doi:10.1001/archneurol.2011.153
2. Henryk Barthel, et al. “Cerebral amyloid-? PET with florbetaben (18F) in patients with Alzheimer’s disease and healthy controls: a multicentre phase 2 diagnostic study.” The Lancet Neurology, May 2011, vol. 10, issue 5, pages 424 – 435, doi:10.1016/S1474-4422(11)70077. Published online April 11, 2011.
3. “To Compare Positron Emission Tomography (PET) Measurements of Fibrillar Amyloid Burden.” www.clinicaltrial.gov/ct2/show/NCT00991419?term=D2750N00006&rank=1. Accessed July 18, 2011.


 May 27, 2026
May 27, 2026 









