GE Healthcares Signa MR750 provides better consistency and improved SAR over previous-generation 3T systems.
New and improved techniques in magnetic resonance imaging (MRI) are popping up every day, stemming from innovations developed by physicians and manufacturers alike. Many of these new techniques are especially conducive to high-field systems — 3.0 Tesla (3T) systems and higher.
3T Advances
While 3T MR imaging has been around for several years, current-generation systems have overecome previous technical roadblocks, enabling the adoption of new techniques. Lawrence Tanenbaum, M.D., FACR, associate professor of radiology, director of MRI, CT and outpatient/advanced imaging development, Mount Sinai School of Medicine in New York, says that MRI manufacturers have done a good job of addressing thorny issues that have plagued earlier 3T scanners. These new systems, including GE Healthcare’s SIGNA MR750, have helped imaging consistency from patient to patient, improving specific absorption rate (SAR) management, as well as management of RF inhomogeneity issues for body applications.
“They’ve done a wonderful job of improving SAR management over their most recent system by setting a SAR model for each individual patient by virtue of a calibration scan,” Dr. Tanenbaum said. “It has essentially eliminated SAR considerations, even for the most demanding exams such as the pelvis, shoulder and hips.”
“The RF inhomogeneity effects are much less perceptible now,” he said. “In the past, we’ve used pads to make the RF environment more homogeneous. On the current system, we no longer use these pads. Even without the use of the pads, the consistency and performance and the homogeneity of the end images is significantly better than it was on the old system with the pads. We’re getting a much better level of image quality.”
Dr. Tanenbaum said some of most exciting new techniques are based on volumetric imaging. Volumetric imaging isn’t new, but had a limited range of contrast available, according to Dr. Tanenbaum. The manufacturers have developed volumetric techniques that have a wider range of contrast available that are fast spin echo-based.
“This allows us to do T2 or FLAIR for the brain and, proton density and proton density FAT SAT for the brain musculoskeletal system,” Dr. Tanenbaum said. “This augments the previously available volumetric techniques for T1 and cisternography. Volumetric techniques are available on 1.5T and 3T, but they synergize with the higher signal-to-noise of a 3T scanner to make thinner slices of high diagnostic quality. For our routine tumor evaluations we will get volumetric T1, T2 and FLAIR, instead of just doing T2 and FLAIR in one or two individual planes. For our MS studies, we’ll get volumetric FLAIR instead of doing multiplanar separate scans. This saves us about 50 percent on time and gives us sub-millimeter voxels to approve sensitivity to early disease.”
Parallel imaging capabilities are also enhanced over the last generation, said Dr. Tanenbaum, allowing radiologists to further leverage the signal-to-noise of 3T.
“Recently developed enhancements in parallel imaging such as ARC have enabled higher levels of acceleration,” Dr. Tanenbaum said. “Multiple directions of parallel imaging further stresses the signal-to-noise equation and better leverages the boost to the signal-to-noise advantage that 3T provides over 1.5T. As a result, scan times have been further reduced for many applications.”
Sodium MRI
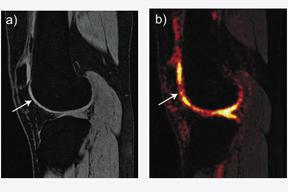
A technique dubbed sodium MRI may help diagnose osteoarthritis in knee joints years before the condition fully develops. While traditional MRI works by exciting hydrogen protons in the body, this technique focuses on a structural material in cartilage, glysosaminoglycan, which occurs in a complex with sodium. Sodium has its own set of excitation and relaxation frequencies and is more restricted to cartilage than water is. Since the element is far less common to the body than hydrogen is, using this scanning technique has generally produced inferior image quality, resolution and signal to noise.
Recent technological innovations have allowed clinicians to develop sodium MRI into a viable clinical scan, according to Gary Gold, M.D., associate professor of radiology at Stanford School of Medicine.
“We thought if we took a new MRI sequence with 3D imaging and short echo times developed by electrical engineers at Stanford, and combined it with a high-quality coil and a 3T scanner, we might be able to get the kind of image resolution that we really need to see cartilage,” said Dr. Gold.
Dr. Gold and other clinicians at Stanford have imaged about 17 patients who tore their anterior cruciate ligament (ACL) in the past 10 years in order to see differences in sodium signals between a patient’s injured knee and a healthy knee.
“The next step is to use this to understand the time course in changes in the proteoglycans in the cartilage after injury and then try interventions — changes in physical therapy, different surgeries, different drug treatments — that might slow or prevent the arthritis from occurring.”
In all cases so far, there has been significant loss of glysosaminoglycan found in the sodium MRI scans within three years of an ACL injury, potentially allowing for accelerated diagnosis.
Dr. Gold and other clinicians at Stanford believe that using a 7T MR scanner will potentially yield better sodium MRI images, as a high field strength is needed for the scan.
Shutter Speed Modeling
Some of the most interesting techniques in MRI involve shutter speed modeling. According to Patrick Bolan, Ph.D., assistant professor, department of radiology, Center for Magnetic Resonance Research, the University of Minnesota, new work on the “shutter speed model” aims to differentiate benign and malignant breast tumors.
“The way dynamic breast contrast-enhanced MRI at 1.5 or 3T is done is by acquiring a series of images before and even after the contrast uptake. The typical way of doing it is one image/per minute over both breasts,” said Bolan. “Aurora Imaging Technology goes with a higher time resolution and a lower spatial resolution, so they might get one every two to three minutes. If you can go faster — with prostate, clinicians often do 10-20 second time resolutions — you can do a good job at characterizing the contrast uptake curve. If you knew the gadolinium concentration with high temporal resolution, you can fit that to a model of what the tissue is really represented by. If you can model that carefully, it’s very useful. You can look at the density of the capillaries and the permeability of the capillaries.”
Charles Springer, Ph.D., and his team at the Oregon Health and Science University (OHSU), are doing work that involves a refined model with more parameters. “Springer did the study with practical time resolutions. Fitting that with his model, he was able to achieve better separation between benign and malignant tumors. He claims the one small change he made in the model was the most sensitive thing that differentiates between the two tumors. They expect this will be more broadly applicable down the road,” said Bolan.
An MRI exam aided by this shutter-speed model analysis may provide a much clearer diagnosis and will become the intermediate step between a mammographic scan and a biopsy intervention, if breast cancer is suspected from both the mammogram and the MRI results, reducing the number of unnecessary biopsies, according to OHSU.


 May 22, 2026
May 22, 2026 









