July 29, 2013 — GE Healthcare has initiated a Class I recall of most of its SPECT nuclear imaging systems and called for its systems to not be operated until the vendor can inspect the scanner. The recall follows the death of a patient after a portion of the scanner fell onto them during the scan on an Infinia Hawkeye 4 system at a VA medical center facility in New York.
On June 17, GE sent an urgent medical device correction letter to all affected customers. The letter identified the affected product, and recommended that qualified service personnel maintain the equipment and that preventative maintenance procedures are executed according to labeling. In addition, users were told that safety chapter sections should be re-reviewed with personnel to ensure proper operation of the equipment.
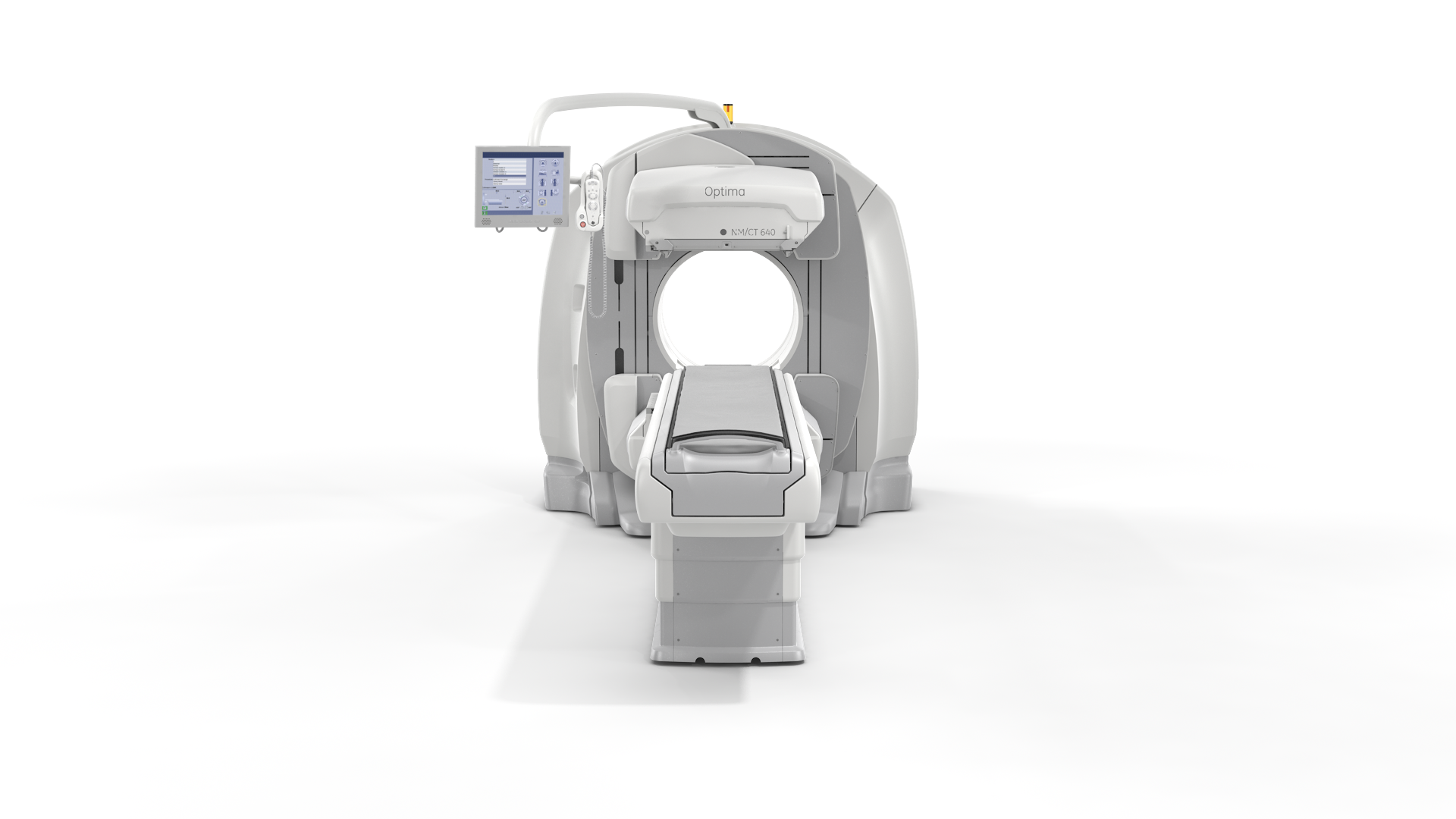
On July 3, GE notified hospitals again via an urgent medical device recall letter and follow-up telephone calls, notifying that it was recalling several more of its nuclear imaging systems. The company said serious injuries or death could occur due to the failure mode associated with the recall. In the second notification, GE included most of its nuclear medicine systems, including the Infinia nuclear medicine systems, VG and VG Hawkeye nuclear medicine systems, Helix nuclear medicine systems, Brivo NM615, Discovery NM630, Optima NM/CT640 and the Discovery NM/CT670.
Healthcare facilities are instructed to cease use of the above mentioned nuclear imaging systems until a GE Healthcare field engineer is able to do a complete inspection of the system and perform any necessary repairs at no cost. GE Healthcare representatives will contact hospitals to arrange for the inspection. If an issue with the support mechanism fasteners is found on a system, the GE field engineer will coordinate the replacement of impacted parts in the gantry to ensure the system is operating safely and meets all specifications.
Investigation of the Accident
In its July 3 recall letter, GE said it was informed of the patient’s death on June 5. GE Healthcare responded and is working with the facility and all appropriate government agencies to complete a thorough investigation.
The company was permitted access to the equipment on June 17. GE Healthcare determined the bolts securing the camera to the gantry were loose, thereby stressing the support mechanism and resulting in the incident. The safety concern is related to a potential patient entrapment or crush hazard if the camera falls during a patient exam, the company said.
Because of the similarities in the design of support mechanisms across many GE products, the company included in its June 17 recall all types and manufacturing dates of Infinia and Infinia Hawkeye nuclear medicine systems; all types and manufacturing dates of VG and VG Hawkeye nuclear medicine systems; and all types and manufacturing dates of Helix nuclear medicine systems by Elscint Ltd. In the July 3 letter the company expanded this recall to include the Brivo NM615, Discovery NM630, Optima NM/CT640 and the Discovery NM/CT670 due to similarities in basic mechanical concepts between these devices and the Infinia systems.
As of Aug. 1, GE reported it had nearly complete its inspections of its SPECT systems installed globally and that systems requiring repair will be returned to service shortly.
GE Healthcare representatives are contacting all known medical facilities that have a GE system to arrange for an inspection. For questions about this recall, contact your GE Healthcare service representative at (800) 437-1171.??


 May 08, 2026
May 08, 2026 









