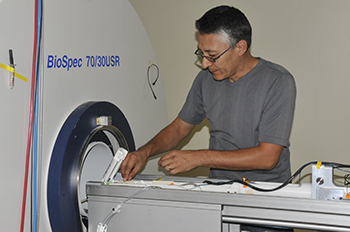
Sergei Liachenko, Ph.D., director of the bio-imaging program, prepares to create an MRI scan of a rat's brain.
September 10, 2014 — Some say a picture is worth a thousand words, but when that picture is used to study a brain, its worth is immeasurable.
Creating such images is the task of Sergei Liachenko, M.D., Ph.D., director of the bio-imaging program at the U.S. Food and Drug Administration’s (FDA) National Center for Toxicological Research (NCTR). Working in a small lab on the NCTR campus south of Little Rock, Ark., Liachenko and his team, in collaboration with the Center for Drug Evaluation and Research (CDER), are studying the effects of various chemicals on a live rat’s brain. Their goal is to prove that studying a live animal with a research-grade magnetic resonance imaging device can be at least as accurate, if not more so, as studies performed under current research procedures.
“We are trying to demonstrate that imaging can be used for testing physical damage to the brain, known as neurotoxicity,” Liachenko says, “and when it comes to studying neurotoxicity, the more detailed a picture we can take, the better.”
That is where magnetic resonance imaging (MRI) comes in. It provides a three-dimensional (3-D) and very sophisticated view of the brain.
How Does MRI Work and What are Its Benefits?
MRI machines use very powerful magnets to bring the water in the body’s cells to a “resonant” state. The machine then sends a resonating radio frequency, which travels through the body and is reflected back. The machine uses the information from this reflection to form a three-dimensional image of the area being scanned. MRI is widely used in hospitals to help identify diseases.
Currently, most scientists look for the damaged brain tissue by sampling multiple pathology slices of the brain of a deceased animal. But Liachenko and his CDER colleague Joseph Hanig are concerned this method could miss some damage because the entire brain is not tested. By contrast, MRI can picture the whole brain in contiguous slices, allowing every part to be examined for changes or damage.
“This assessment allows the entire brain to be scanned and produced in a 3-D representation, so we can view the tissues in different projections, and we don’t need to worry about missing potential damage because it’s in an area that was not examined,” says Liachenko.
Another benefit to using an MRI device is the ability to test a single rodent efficiently, repeatedly and safely. “It’s just a magnetic field, and we’re using what’s considered the safest imaging modality. That’s why we can put the same animal in the machine many times,” Liachenko says.
Studying the same animal over a long period of time, Hanig adds, “gives us more data that’s arguably more accurate than data from multiple subjects.” In addition, these MRI assessments are similar to those made in humans and therefore they could be connected with clinical research.
Yet another important advantage of using the MRI is that the imaging takes hours to complete and analyze, while conventional histological analysis procedures take weeks. This change in timeframe, along with fewer subjects being used, could make imaging a more cost effective option.
Imaging at FDA
MRI is used all over the globe to investigate live animals, but what is unique to NCTR, in collaboration with CDER, is its study to develop MRI biomarkers of neurotoxicity. By using a set of compounds that cause known damage to the brain, the researchers can check the efficiency of the MRI scans compared to the conventional methods by making sure the MRI machine finds the same problems, and perhaps even more.
“Should the biomarkers prove to be reliable and the qualification is met, it will send a huge message to the pharmaceutical community that this method is good for assessing for potential brain damage,” Liachenko says. “In our view, this MRI research will create the opportunity to provide much safer drugs to people, and that’s the reason for our work.”
This article appears on FDA’s Consumer Updates page, which features the latest on all FDA-regulated products.
For more information: www.fda.gov/ForConsumers/ConsumerUpdates/ucm412293.htm?source=govdelive…


 May 22, 2026
May 22, 2026 









