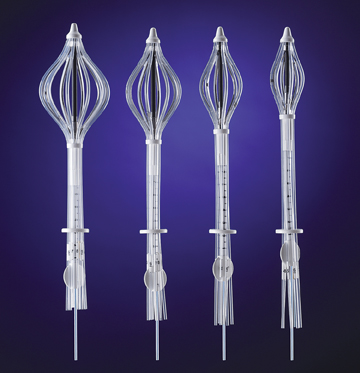
The SAVI applicator has a strut-based, open architecture design that precisely targets radiation where it is needed most, which
Today, only 12 percent of breast cancers in the United States are treated with accelerated partial breast irradiation (APBI). Breast brachytherapy is a form of APBI that delivers radiation dose from within the breast and tumor cavity using a tiny radioactive pellet of iridium-192. The clinical benefits of this targeted approach to radiation delivery are clear — reduced radiation exposure to healthy tissue, better cosmetic results and fewer side effects, and allowing more future treatment options in the event of a recurrence or new primary cancer.
Despite these advantages, breast brachytherapy adoption has slowed in recent years, often in favor of other accelerated treatment options. While this has led some to question the importance of breast brachytherapy and whether it will stand the test of time among early-stage breast cancer treatments, I would argue it’s a standard of care that physicians should feel obligated to offer to every eligible patient.
Is breast brachytherapy underutilized? Absolutely. Is it relevant? More than ever — and it’s here to stay.
Evolution of Breast Brachytherapy
Modern breast brachytherapy was first introduced in 1991, when a patient requested a treatment that was appropriate not just for her cancer, but for her busy lifestyle as well. The results of the five-day treatment were impressive, and led to a number of clinical trials to compare it to the current standard of care, six weeks of whole breast irradiation (WBI). Though the benefit of a shorter course of treatment was obvious — enabling women to return to their normal family and work schedules after just five days — breast brachytherapy gave physicians the opportunity to explore an idea many of us had been pondering for awhile: if most recurrences happen in the same location as the original cancer, why were we treating the entire breast with radiation? The rationale behind breast brachytherapy was to intensify treatment to the area most at risk for recurrence, treating just the affected part of the breast in order to control the cancer, reduce side effects and improve overall quality of life.
For the first decade, the only form of breast brachytherapy was interstitial brachytherapy, which uses multiple catheters to deliver radiation to a two-centimeter margin beyond the lumpectomy cavity. Despite excellent outcomes and promising clinical trials, interstitial proved to be too skill-dependent for widespread adoption. Intracavitary brachytherapy was introduced in 2002 when the U.S. Food and Drug Administration (FDA) approved the MammoSite balloon applicator — the first single-entry applicator for the delivery of breast brachytherapy. The ease of use of the balloon led to a rise in the popularity of breast brachytherapy in the United States.
While five-year results released from the American Society of Breast Surgeons (ASBS) showed excellent local control with balloon brachytherapy, they also underscored the dosimetric limitations of a single-channel device. The inability to shape the radiation dose poses a greater risk to healthy tissue and led to higher rates of toxicities, including seroma, fat necrosis and skin changes.
In 2006, the FDA approved the SAVI applicator from Cianna Medical, which combined the dosimetric advantages of interstitial brachytherapy with the simplicity of a single-entry balloon. The strut-based, open architecture design of SAVI provides physicians with greater control of the radiation dose, enabling precise targeting of the tumor bed while avoiding critical structures such as the skin, chest wall, heart and lungs. With the ability to customize radiation based on patient-specific anatomy, the SAVI technique increases the number of women who may be eligible for breast brachytherapy. The introduction of SAVI was a significant advance in breast brachytherapy, and it’s a tool every radiation oncologist should have in his or her toolbox.
The Rise of Accelerated Radiation Options
Across the country and throughout the world, patients are seeking alternatives to six weeks of WBI that allow them to maintain their health and return to their normal lives as soon as possible. There are a number of radiation options that offer accelerated treatment schedules for lower risk cancers. While patients deserve options, we must carefully examine these technologies and the evidence behind them to determine where they fit in the treatment paradigm.
Intraoperative Radiation Therapy — Too Little? Intraoperative radiation therapy (IORT) is a method of APBI that reduces radiation delivery to just one intraoperative treatment. The convenience of a “one-and-done” radiation treatment is undeniable for patients, which is presumably why some institutions have adopted IORT as part of their breast cancer programs prior to having long-term data to support its efficacy.
However, convenience should not outweigh efficacy and safety, and data are beginning to indicate that IORT is associated with inferior local control and higher toxicities. Last fall, results from two Phase III trials comparing IORT to WBI were published, and both reported significantly higher local recurrence rates with IORT — 2.5 times higher in the TARGIT-A trial and 11 times higher in the ELIOT trial (absolute differences were 2 and 4 percent greater, respectively).
In both studies, the whole breast radiation arm was better than the IORT arm. The fact that these differences seem to be increasing over time is even more concerning — how many local failures will there be at 10 years? In addition, the ELIOT trial reported a high level of fat necrosis (17 percent) with IORT at five years, which could mean that normal breast tissues have trouble surviving a single, very large dose of radiation.
There are serious limitations of IORT, including a lack of pathology, image guidance and quality control. Since radiation is delivered at the time of lumpectomy, physicians are making treatment decisions without complete pathologic data. The lack of information regarding margins, nodal status and so on means physicians are irradiating the patient without even knowing if they are a candidate for ABPI.
There’s also no assurance the target tissue is being sufficiently covered. With no image guidance and a lack of quality control, it is impossible to confidently identify and document where dose is delivered relative to the cavity, as well as the skin, chest wall and other critical structures. Thus, the overall quality of treatment remains unknown and likely varies from one institution to the next, especially with the intraoperative electron beam method.
This variation is compounded by the insufficient dosimetry of techniques that deliver 20-21 Gy in a single fraction. This dose distribution is a dramatic departure from standard radiobiological principles, which cannot be applied at absolute doses of greater than 8 Gy. As a result, a massive dose is delivered to the cavity walls, putting the patient at risk for long-term complications such as fibrosis. With the Zeiss Intrabeam (the IORT method used in the TARGIT-A trial), as little as 5 Gy is being delivered at 1 cm beyond the cavity, which is an inadequate dose to control cancer. In contrast, brachytherapy delivers 3.4 to 4 Gy for each fraction.
Hypofractionated Whole Breast Irradiation — Too Much? In the past several years, randomized clinical trials in Canada and the United Kingdom have compared outcomes of hypofractionated whole breast irradiation (hWBI) to conventional WBI. While there are a variety of hWBI fractionation schedules currently in use, the most common involves the delivery of 42.5 Gy over 16 fractions, also known as “Canadian fractionation.” Depending on whether a boost is required, this results in a three- or four-week treatment.
Compared to standard WBI, the randomized trials for hWBI have shown little difference in local control and survival for lower risk cancers. However, there is uncertainty regarding patient selection and whether these results can be applied to all patient subgroups. Further research is needed to answer questions regarding the role of boost radiotherapy and the suitability of hWBI for young women, high-grade disease, close/positive margins, large breasts or those receiving chemotherapy.
Other concerns regarding hWBI involve late toxicities and cosmesis. We know from published literature that a high integral dose (prescribed dose times the volume treated) and high dose per fraction lead to late complications that may not present themselves until 10-15 years post-treatment, including fibrosis, skin changes, rib fractures and other long-term side effects. Both the integral dose and dose per fraction are much higher with hWBI than either standard WBI or brachytherapy. In addition, the hWBI trials have not assessed the long-term risk of cardiac toxicity for left-sided cancers, so we must continue to be vigilant for these late effects as follow-up continues.
Some patients may request hWBI as a non-invasive alternative to brachytherapy, but reducing the length of treatment from six to four weeks is not as attractive as six weeks to five days. We must remember that hWBI still irradiates the entire breast, which abandons the initial premise that makes breast brachytherapy so attractive: reducing the overall target size to the area most at risk in order to minimize unnecessary radiation, reduce side effects and improve quality of life.
Quality of life is also impacted by length of treatment. In my opinion, the lifestyle benefit of an accelerated treatment is somewhat lost with hWBI, especially if a boost is required.
Breast Brachytherapy — Just Right? So here we have two accelerated treatment options for treating lower risk cancers: IORT, which is incredibly convenient, but doesn’t appear to be as effective in controlling cancer; and hWBI, which is effective, but still exposes healthy tissue and critical structures to radiation while offering only a slight reduction in overall treatment time relative to standard WBI.
I would argue that breast brachytherapy offers the best of both worlds, and thus remains a very attractive option for certain early-stage breast cancers. It offers equivalent tumor control, superior cosmesis and lower toxicity, and at five days it’s still far more accommodating to women’s lifestyles than WBI (either standard or hypofractionated schedules).
There is a tremendous body of clinical research supporting the use of breast brachytherapy, which is why it has been accepted and endorsed by major medical societies including the American Society for Radiation Oncology, ASBS and the American Brachytherapy Society. The scientific evidence supporting brachytherapy is robust and mature:
1. A Phase II RTOG trial with 12.5 years of follow-up shows extraordinarily low recurrence rates. This is particularly meaningful given that the study included many patients with high risk factors for recurrence, including positive nodes, grade 3 tumors, ER negative tumors and patients under 50 years of age.
2. Two matched pair analyses have reported identical rates of local and regional recurrence at 10 years when comparing interstitial brachytherapy and WBI.
3. A randomized European trial with more than 10 years of follow-up showed equivalent local control and significantly better cosmetic outcomes for breast brachytherapy.
The Future of Breast Brachytherapy
For those still on the fence regarding the five-day radiation treatment, the definitive answer will come from two large randomized trials. The results of NSABP B-39/RTOG 0413, a randomized 4,300-patient Phase III trial comparing five-day APBI to six-week WBI, are expected in a couple years. As a co-principal investigator of the National Cancer Institute-sponsored study, I believe the results — if they demonstrate the equivalent control and low toxicities seen in other large scale studies — will put to rest any lingering questions regarding the suitability of breast brachytherapy for properly selected patients.
The trial closed to new patients in April 2013, and the data will be released after the last patients have been followed for a sufficient time to allow for failures to occur. With median follow-up between five to seven years, there have been no meaningful statistical differences between the two arms that would have required the statisticians to release the data sooner, which is a good indication for APBI.
Similarly, the European GEC-ESTRO Phase III trial comparing brachytherapy to WBI is complete. One-year toxicity results — illustrating superior cosmesis with brachytherapy — were reported in 2013, and recurrence data are expected in 2015.
Although the NSABP B-39 and GEC-ESTRO results will be meaningful, I don’t believe it’s necessary for physicians to delay implementation of breast brachytherapy until outcomes are available. To do so ignores the large body of evidence supporting the safety and efficacy of brachytherapy. In addition, there are many examples in medicine where treatments or procedures have come into practice prior to the availability of Phase III data, including breast conservation therapy, sentinel node mapping and prostate brachytherapy.
Meanwhile, there are a number of other breast brachytherapy studies underway, including 12 randomized trials in progress. One of the largest brachytherapy studies is expected to be published this year. With more than 1,500 patients from six institutions over 20 years, the PROMIS study will provide long-term evidence supporting interstitial brachytherapy. The SAVI Collaborative Research Group, which studies the long-term outcomes of strut-based brachytherapy, continually reports exceptional results (excellent local control and very low toxicities) from a database of more than 1,000 patients from 14 institutions. Researchers are expected to release five-year data this fall.
Patients need to know there are many options for treating early-stage breast cancer, and brachytherapy is an ideal choice for certain patients. Based on the current data, and the potential impact of forthcoming studies, I predict that in the next five years, as much as 40 percent of breast cancers in the United States will be treated with breast brachytherapy.
While breast brachytherapy may be underutilized, overwhelming evidence supports it as a new standard of care for many women. It’s effective, safe and reduces the burden that breast cancer treatment places on women and their families.
Robert R. Kuske, M.D., FAACE, is a radiation oncologist at the Arizona Breast Cancer Specialists in Scottsdale, Ariz. Passionate about helping women with breast cancer, he pioneered “accelerated partial breast irradiation,” a five-day alternative to the conventional six weeks of external beam whole breast irradiation in select early-stage breast cancer. He is a co-principal investigator of the North American 4,214-patient Phase III trial through the National Surgical Adjuvant Breast and Bowel Project and Radiation Therapy Oncology Group, approved and funded by the National Cancer Institute. He has treated more than 2,000 women with brachytherapy over his 30 year career.


 May 07, 2026
May 07, 2026 









