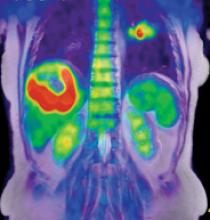
The addition of magnetic resonance (MR) imaging and spectroscopy to positron emission tomography (PET) is more expensive and more technically challenging compared with PET/computed tomography (CT). PET/CT is successful because the inclusion of CT has major advantages: accurate lesion localization, the identification of non-PET avid lesions and effective attenuation correction in a rapid, efficient combined examination. The addition of CT is particularly valuable for lungs and liver, where fluorodeoxyglucose (FDG) PET is limited by spatial resolution and relatively low target-to-background differential biodistribution. Presumably, PET/MR may disclose unique important diagnostic and prognostic information in selected patient groups.