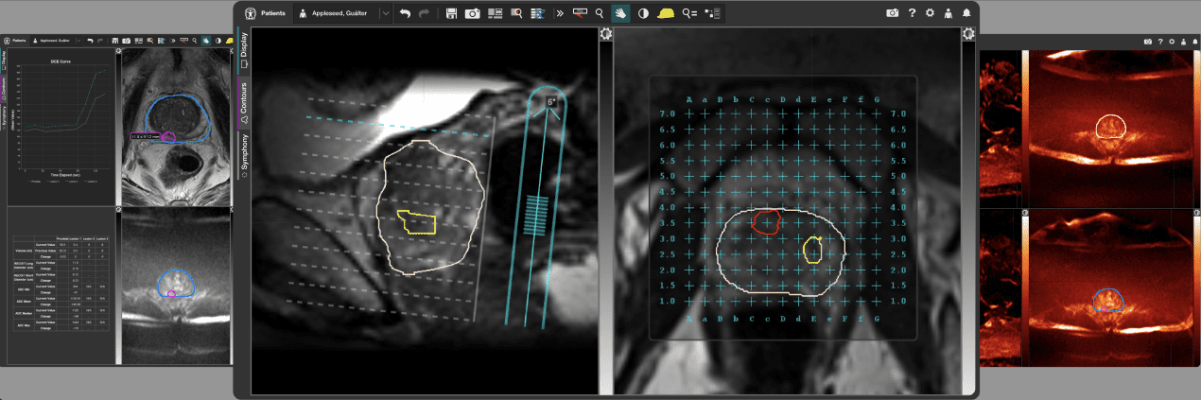
July 11, 2024 — GE HealthCare’s MIM Software, a global provider of medical imaging analysis and artificial intelligence (AI) solutions, is excited to introduce MIM Symphony HDR Prostate to support high dose-rate (HDR) brachytherapy. This innovative solution aims to increase clinician confidence and help improve patient outcomes by providing direct tumor visualization from magnetic resonance imaging (MRI) scans during live ultrasound procedures for HDR prostate treatments.
MIM Symphony HDR Prostate stands out in the market by correcting MRI orientation and offering MRI guidance during HDR prostate procedures.
HDR brachytherapy is a prostate cancer treatment that involves implanting radioactive sources into the prostate gland via needles. Typically, clinicians use computed tomography (CT) or ultrasound imaging for planning these procedures. Incorporating MRI into brachytherapy treatment planning can reduce toxicity to critical structures.[ii] However, the challenge of accurately aligning MRI contours with live ultrasound images has hindered the use of MRI in HDR treatments.
Dr. Peter Rossi, MD, FABS, Chairman of the Board at the American Brachytherapy Society and Radiation Oncologist at Valley View Hospital, said, “I often get the question, what is the best imaging modality for planning HDR prostate procedures, ultrasound or CT? My response is always MRI. MRI guidance enables more confident, precise treatments and reduces toxicity to organs at risk. I’ve had the pleasure of providing my patients with precise treatments using MRI guidance in MIM Symphony HDR Prostate, and I look forward to other clinics having the same opportunity.”
GE HealthCare’s MIM Software developed MIM Symphony HDR Prostate to align contours on preoperatively acquired MRI with live ultrasound, providing clear visibility of the prostate, lesions, and critical structures during HDR prostate procedures. Aligning the MRI contours with the ultrasound assists clinicians in precise needle placement by:
· Defining the lesion and tracking changes to guide needle placement, even as the prostate moves during the procedure;
· Correcting differences between MRI supine and ultrasound lithotomy orientations with ReSlicer, a tool in MIM Symphony HDR Prostate; and
· Automatically digitizing needles on CT or ultrasound planning images as well as performing needle review and free length checks.
“Our goal with this solution is to empower clinicians to plan and deliver safe, more informed, and precise treatment for patients with prostate disease,” said Andrew Nelson, CEO of MIM Software, GE HealthCare. “MIM Symphony HDR Prostate exemplifies decades of MIM Software and GE HealthCare innovations that enable precision care, and we’re thrilled to expand our impact in brachytherapy.”
This new solution is the latest addition to GE HealthCare’s MIM Software portfolio of vendor-neutral solutions for radiation oncology. Following the recent acquisition of MIM Software’s portfolio – including MIM Maestro, Contour ProtégéAI+, and MIM Symphony LDR – GE HealthCare continues to offer a broad portfolio of solutions across precision imaging and radiopharmaceuticals as well as seamless interoperability of radiation oncology departmental systems to drive efficiency and optimize care across care pathways.
For more information: https://www.gehealthcare.com/
Find more AAPM24 conference coverage here
References:
i] MIM Symphony HDR Prostate is commercially available in the U.S. and CE marked. Not available for sale in all regions. Not commercially available in all markets.
[ii] Register SP, Kudchadker RJ, Levy LB, et al. An MRI-based dose--response analysis of urinary sphincter dose and urinary morbidity after brachytherapy for prostate cancer in a phase II prospective trial. Brachytherapy. 2013;12(3):210-216. doi:10.1016/j.brachy.2012.10.006


 May 12, 2026
May 12, 2026 









