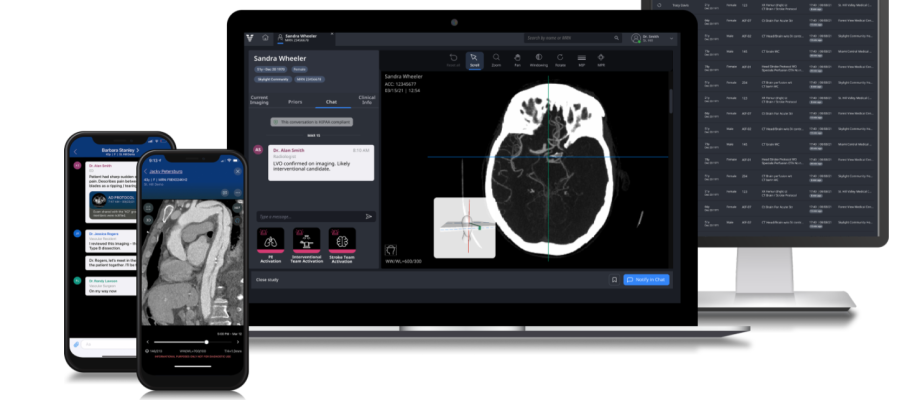
February 23, 2022 — Viz.ai, the world leader in artificial intelligence (AI) powered care coordination, announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for Viz Aneurysm. The new algorithm uses AI to detect suspected cerebral aneurysms, enabling hospital systems to ensure that once detected, patients are captured and the aneurysm workflow across an entire health system is standardized. This ensures that patients receive the appropriate follow-up care and creates significant financial benefits for hospital systems and payers. Viz Aneurysm will serve as a first of its kind population health tool to facilitate population screening and enhanced care management.
Today, detecting cerebral aneurysms and coordinating care for patients with this condition is challenging for non-specialist centers, and care pathways can be complex. If left untreated, an aneurysm can rupture, spilling blood into the surrounding tissue, which causes a life-threatening and costly emergency called a subarachnoid hemorrhage. According to Etminan et al, approximately three percent of the population has a cerebral aneurysm and approximately 30,000 people suffer subarachnoid hemorrhage in the U.S. every year, which is associated with a 40 percent mortality rate.
A study at the University of Toronto tested the accuracy of Viz Aneurysm using 528 CTAs with 674 aneurysms broadly distributed across the cerebral vascular territories, encompassing anterior and posterior circulation. Data analysis demonstrated 94% accuracy for the algorithm.
“[Aneurysms] can be often missed because they require a very methodical diagnostic approach. The Viz.ai aneurysm algorithm was able to detect cerebral aneurysms in consecutive CTAs. The model has demonstrated that a deep learning AI algorithm can achieve clinically useful levels of accuracy for clinical decision support and will help us to improve how we help aneurysm patients,” concluded Dr. Vitor Mendes Pereira, director of Endovascular Research and Innovation, University of Toronto.
“Viz Aneurysm has the potential to significantly increase the number of aneurysms detected and clinically followed,” said Jayme Strauss, chief clinical officer, Viz.ai. “The combination of detection with an ability to schedule patients for neurovascular specialist follow-up is an important advancement for aneurysm patients, helping patients obtain the necessary follow-up from this potentially deadly disease and driving improved outcomes on the population health level.”
The Viz Aneurysm module is additive to the Viz Intelligent Care Coordination Platform, which is clinically validated and reimbursed by Medicare and proven to save time, and improve patient outcomes and access to care. Viz is the most comprehensive and effective neurovascular and vascular AI platform that is already helping to coordinate care across a broad list of clinical applications, including ischemic and hemorrhagic stroke, pulmonary embolism and aortic disease in more than 1,000 hospitals.
For more information: https://www.viz.ai/


 May 12, 2026
May 12, 2026 









