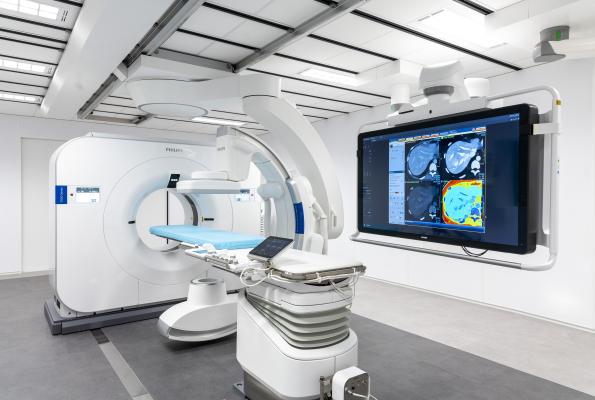
November 28, 2021 — Royal Philips, a leader in health technology, unveiled its vision for interventional medicine by bringing its breakthrough spectral CT imaging technology into a hybrid Angio CT suite. By combining the company’s Spectral CT 7500 system and its image-guided therapy system – Azurion with FlexArm in a single interventional suite solution, Philips aims to give interventionalists immediate table-side access to these two key imaging modalities so they can perform procedures requiring both CT and angio guidance in one room. For the development of this integrated solution Philips is closely collaborating with leading clinical institutes such as Mayo Clinic and Baptist Health’s Miami Cardiac & Vascular Institute. It is being presented at the 2021 Radiological Society of North America Annual Meeting, Nov. 28 – Dec. 2 in Chicago.
The goal of combining the breakthrough, first of a kind Spectral CT 7500 with the Azurion platform is to provide a seamless workflow environment in which all the necessary imaging modalities - are available directly at the patient table, fully controllable from the table-side work spot. When not in use, each modality glides smoothly away from the patient table into a parking position, giving the interventional team unrestricted access to the patient. Each system can work stand-alone if required. For interventionalists, it enables seamless efficient workflows without the need to move the patient from one imaging suite to another. For patients, this means their physicians can leverage the strengths of both systems during a single procedure.
Requiring just one scan to capture all the spectral information required to differentiate and quantify different tissues, Philips’ always-on spectral CT technology enables improved detection, delineation, and quantification of lesions, leading to better-informed planning for minimally-invasive procedures and more precise interventions. In oncology, spectral CT has also demonstrated a higher sensitivity in detecting malignant findings and has improved readings of incidental findings.1, 2
“There has been a tremendous evolution in the image-guided therapy environment,” said Dr. Barry T. Katzen, founder and chief medical executive of Baptist’s Health’s Miami Cardiac & Vascular Institute. “We are really excited about the merger and integration of these technologies in that it might allow us to look at the treatment of a number of diseases in very new and innovative ways, potentially offering new treatment opportunities and improving patient care. I am looking forward to investigating the possibilities of this promising innovation.”
“With this innovation, we are taking a uniquely patient-centric approach, aimed at bringing the latest interventional imaging modalities to the patient table rather than moving the patient to the individual modalities,” said Karim Boussebaa, general manager of image guided therapy systems at Philips. “We are excited to be co-creating the future of image guided therapy with leading clinical partners by combining the best of our award-winning modalities into a single suite.”
Join Philips at RSNA 2021 where the company will spotlight its latest advanced technology driving connected workflows and smart connected imaging systems.
For more information: www.usa.philips.com
References:
1 Analysis by Aarhus University Hospital Aarhus, Denmark. Results from case studies are not predictive of results in other cases. Results in other cases may vary.
2 Analysis by University Hospital Cleveland, USA. Results from case studies are not predictive of results in other cases. Results in other cases may vary.


 May 19, 2026
May 19, 2026 









