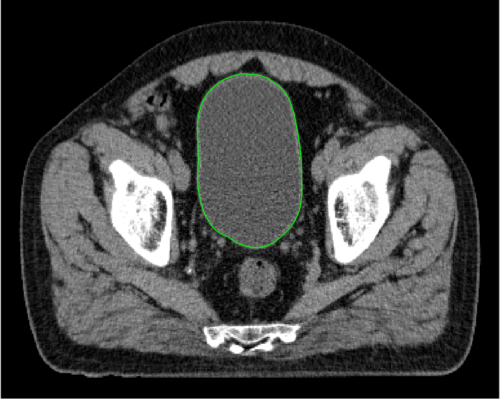
June 9, 2020 — Mirada Medical, a global leader in artificial intelligence (AI) software for the treatment of cancer, has launched an online version of its autocontouring software, DLCExpert, which is now available for cancer treatment centers to use free of charge until July 31, 2020. DLCExpert is one of the world’s first commercially available AI-powered autocontouring software for cancer treatment planning and the only system with over one year of clinically proven use.
Available online, oncologists and dosimetrists can securely upload their treatment planning computed tomography (CT) scans to be autocontoured by DLCOnline, giving them back precious time to focus on patient care. To date, more than 50 cancer centres worldwide have registered for the service.
Hugh Bettesworth, CEO of Mirada Medical said, “Healthcare is Mirada’s business and we have been asking ourselves what more we can do to support cancer care during this crisis, DLCOnline has been created to help clinicians clear the backlog of cases as treatment resumes. By offering the service in all territories where we have regulatory clearance, and free of all charges until the 31st July, we hope to support cancer centres deliver more life-saving patient care.”
DLCExpert is a new product developed using advanced AI technology, and has been cleared for clinical use in the USA, EEA, UK, and India. Last year approximately 16,000 radiation treatment plans were made using Mirada Medical’s autocontouring. DLCExpert has been shown to deliver high quality, consistent organ-at-risk contours while delivering significant time savings when compared with the manual process.
For more information: www.mirada-medical.com
-


 May 27, 2026
May 27, 2026 









