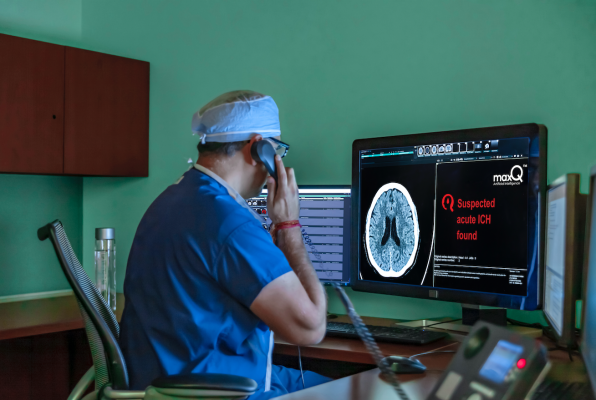
December 16, 2019 — MaxQ AI, a company focused on developing artificial intelligence (AI) applications to enable doctors to reach faster, more accurate decisions when diagnosing stroke, traumatic brain injury, and other life-threatening conditions, has selected Flywheel to support data management and curation and gain efficiencies in their machine learning workflow.
“We selected Flywheel for our machine learning data management due to their unique strengths in medical imaging and research workflows,” said Tom Neufelder, chief technology officer of MaxQ. “With Flywheel we can streamline capture, quality control, and curation of data from our research institution partners enabling us to focus on innovation and development of high-value solutions for our customers.”
Leading research institutions and AI developers are turning to Flywheel for imaging research data management and machine learning applications. Flywheel has recently introduced a number of features aimed at improving machine learning workflows:
Image Annotation Workflows to support labeling of specific image features including support for blind multi-reader studies.
Advanced Multimodal Search to streamline the process of finding data to support ML training, including the ability to search image annotations.
ML Training Sets to simplify training workflows and related documentation to support reproducibility and FDA approvals.
For more information: www.maxq.ai


 May 27, 2026
May 27, 2026 









