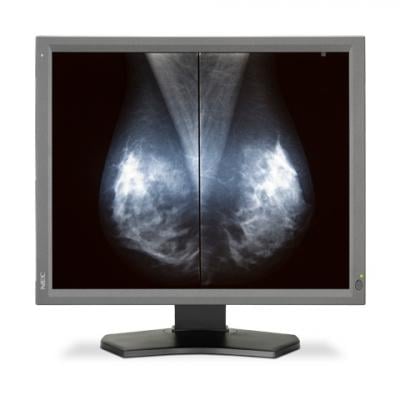
March 3, 2016 — NEC Display Solutions of America announced U.S. Food and Drug Administration clearance of its MultiSync MD211G5 medical-grade monitor for use in tomosynthesis applications and full field digital mammography (FFDM). The 21-inch NEC display is built for the displaying and viewing of digital images for diagnosis by trained physicians.
The 5-megapixel grayscale monitor, which also has been cleared for standard mammography, offers out-of-the-box factory DICOM calibration and uniformity control for consistent imaging across the entire screen. Its IPS panel features a 1200:1 contrast ratio and 500 cd/m2 of calibrated brightness. The MD211G5 display also comes with GammaCompMD QA software for managing conformance to the AAPM TG-18 standard.
Digital breast tomosynthesis (DBT), which combines tomography with digital radiography, renders breast images in 3-D and accounts for more than one-half of the breast-imaging market, according to a report by iData Research.
A separate study by the University of Pennsylvania’s Perelman School of Medicine published in JAMA Oncology found that initial DBT screening led to, among other things, fewer patients called back for sometimes unnecessary imaging.
For more information: www.necdisplay.com


 May 07, 2026
May 07, 2026 









