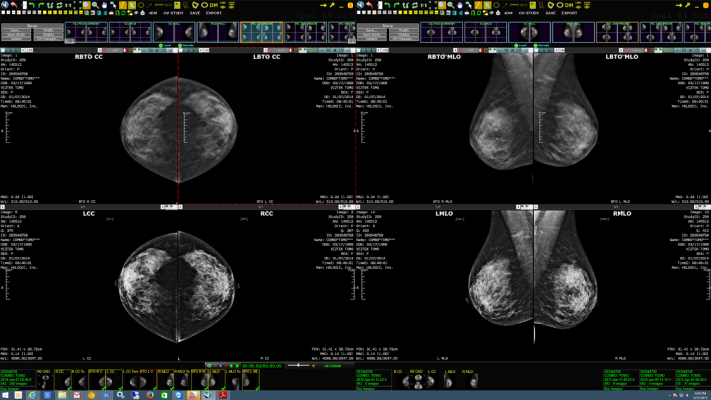
Image courtesy of Viztek
November 24, 2015 — Viztek, a Konica Minolta Company, will showcase the Exa Mammo Viewer – a PACS-neutral, breast imaging management workstation with full diagnostic quality for breast imaging – during the upcoming Radiological Society of North America (RSNA) scientific assembly and annual meeting, Nov. 29- Dec. 3, 2015. Viztek debuted the Exa Mammo Viewer earlier this year as the latest innovation to its Exa platform of healthcare information technology (IT) software for image viewing and sharing.
The Exa Mammo Viewer can be used in conjunction with any facility’s current picture archiving and communication system (PACS), benefiting physicians by enabling comparison with images and reference to patient files for breast ultrasound, magnetic resonance (MR), computed tomography (CT) and more. It is the first and only solution, according to the company, to enable diagnostic quality viewing of any breast imaging study – including digital breast tomosynthesis (DBT) – from any remote location, regardless of the PACS being used. This ability to read, diagnose and compare with other breast imaging studies from any location within the department or offsite creates tremendous efficiencies in the patient care process.
The Exa Mammo Viewer is an extension of the Exa platform. The speed at which radiologists and other physicians can access all breast imaging studies of any size, whether physically located within the department or outside at another reading location, is a result of true zero footprint (ZFP) viewing and server-side rendering (SSR). There is no pre-fetching required and there are no plug-ins required. Full functionality and instant access is available regardless of the operating system. So not only is the hospital IT network freed from the burden of downloading numerous large image files, but regardless of the computer preferences of physicians – MACs, PCs, iOS or Android tablets and phones – all are universally supported. In short, the Exa platform provides fast access anywhere.
For more information: www.viztek.net


 May 22, 2026
May 22, 2026 









