July 22, 2015 - CEIA USA Ltd. announced the availability of the CEIA Healthcare Detection Systems metal detection technology for magnetic resonance imaging (MRI) safety screening.
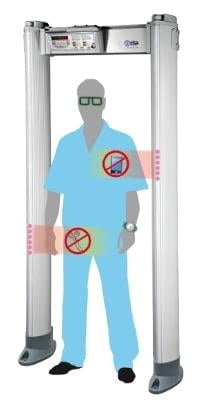
The CEIA MetalMag walk-through detector and CEIA PD240CH handheld detector combine the benefits of both cutting-edge metal detection and ferromagnetic detection functionality to automatically filter and suppress alarms for non-threat items, without compromising the required threat item detection capabilities. As a result, the new systems end false alarm fatigue and guesswork.
MRI safety screening is essential for both patients and staff and should be simple, fast and highly effective every time. The MetalMag walk-through detector automatically detects a wide variety of metallic items and ferromagnetic material threat items, such as scissors, that are considered dangerous in MRI environments, all while eliminating unnecessary false alarms for non-threat items such as shoes, eyeglasses and even underwire bras. The MetalMag features CEIA's precision multi-zone targeting indication to pinpoint the exact location of the dangerous item on the person or in the body of the individual in transit. As a freestanding unit it does not require any specialized installation and can be quickly deployed prior to the MRI screening control point in any facility.
The PD240CH handheld detector provides precise screening and uniform detection of smaller dangerous items for patients prior to being admitted to the MRI magnet room. The portable metal detector combines high sensitivity to ferromagnetic metal masses with immunity to non-ferromagnetic metal masses such as dental fillings, as well as dental and metal limb prosthesis. Additionally, by quickly changing the operational mode, it can become a complete metal detector capable of detecting any type of metal material to include pacemakers, prostheses and vital support devices, as well as any other object carried by the patient that should not be taken into the strong magnetic field area. This mode can also be used to identify, for example, the presence of metal slivers in a patient following an accident or swallowing metal objects.
These solutions were developed in collaboration with Aegys, creator of product and process solutions that enhance MRI safety and improve efficiency.
Other key features of CEIA MRI Safety Screening Systems include:
- Multiple alarm tones and volume setting options for staff-friendly environment
- Broad range of accessories includes optional remote detector network management
Both devices were introduced at the Association for Medical Imaging Management (AHRA) 2015 annual meeting.
For more information: www.ceia-usa.com


 May 20, 2026
May 20, 2026 









