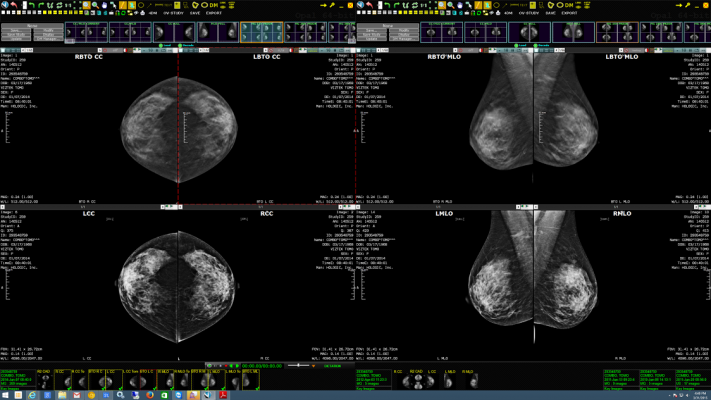
May 5, 2015 — Viztek will showcase its new Exa Mammo Viewer during this year’s Society for Imaging Informatics in Medicine (SIIM) conference, May 28-30 in Washington, D.C.
The advanced imaging solution provides two significant benefits:
- It displays full diagnostic-quality images from any modality, including complete support of digital breast tomosynthesis (DBT); and
- It enables radiologists to read, diagnose and compare breast images while working remotely.
The Exa Mammo Viewer can be used in conjunction with any facility’s current picture archiving and communication system (PACS), enabling comparison with images and reference to patient files for breast ultrasound, magnetic resonance (MR), computed tomography (CT) and more. Physicians benefit from seamless work and fast image download times, and IT benefits from unique back-end efficiencies that are realized from Viztek’s IT –enabled development.
For more information: www.viztek.net


 May 22, 2026
May 22, 2026 









