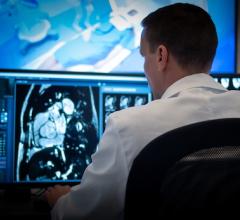
Open Inventor SDK is platform for developers of all types of visualization software on desktop and mobile environments: 2-D and 3-D imaging, image processing and analysis, PACS, diagnosis and surgical planning, device modeling and printing, simulation and training.
The Open Inventor object-oriented API is designed to quickly integrate powerful new visualization capabilities into software applications in a simple and consistent way. It provides comprehensive capabilities for interactive volume visualization, blending, clipping and editing; advanced 2-D and 3-D image processing, analysis, segmentation and registration; and remote 3-D interaction and visualization for Web-based and mobile applications.
For more information: www.open-inventor.com
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now


 April 28, 2026
April 28, 2026