January 13, 2010 – Research validates the efficacy of the recent reclassification of the T category in the International Association for the Study of Lung Cancer’s (IASLC) "Staging Manual in Thoracic Oncology" released in the January 2010 issue of the Journal of Thoracic Oncology.
In the sixth edition of the tumor, node and metastatis (TNM) staging system, there were no changes. Thus, the change in the seventh edition is significant as it is the first revision of TMN classification in lung cancer for 12 years, and represents the most drastic change there has been in the past 30 years.
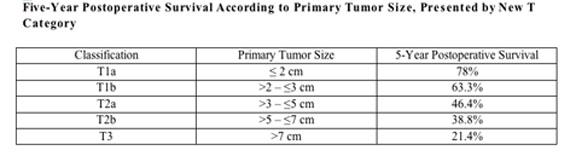
The medical records of 1,393 patients with non-small cell lung cancer who underwent complete resection were thoroughly reviewed for pathologic findings and survival. The stage of all the patients was pathologically defined according to the present international staging system, previously revised in 1997. This study examined the relationship between the cut-off points of the tumor size for the new T category and the pathologic invasiveness
The TNM staging system is useful for both the clinical assessment of tumor progression and the determination of treatment plan, such as surgery, chemotherapy and radiation. In the forthcoming TNM classification, a revision with respect to lung cancer has been proposed by the International Association for the Study of Lung Cancer. The major revision is the T category, which makes an especially strict division by detailed cut points of tumor size (e.g., 2-, 3-, 5- and 7-cm). The proposed T revision has been determined and validated based on the overall survival date from a large international database.
The lead study author, Tokujiro Yano, M.D., called the new T classification, which is based mainly on the tumor size, “appropriate for the pathologic findings of the primary tumor.” The significant differences were observed among newly revised T subsets in at least one incidence of pathologic invasiveness, including lymphatic, vascular, or pleural invasion.
Past chair and author of the IASLC’s staging guide, Peter Goldstraw, added that IASLC will play a central role in the formation of future classification of TMN.
For more information: www.iaslcpubs.org/IASLC-Manual-Thoracic-print.html


 May 06, 2026
May 06, 2026 









