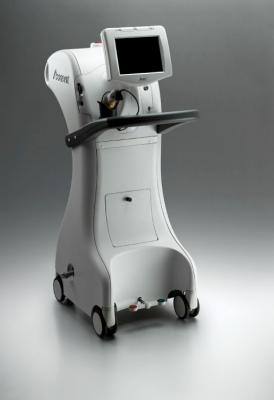
March 15, 2017 — iCAD Inc. announced that more than 3,000 early-stage breast cancer patients have been treated with intraoperative radiation therapy (IORT) using the Xoft Axxent Electronic Brachytherapy (eBx) System. The Xoft System and its suite of products were showcased in the Xoft booth during Physicians’ Education Resource (PER)’s 34th Annual Miami Breast Cancer Conference at the Fontainebleau Miami Beach Hotel in Miami Beach, Fla., March 9-12, 2017.
IORT is a procedure that allows radiation oncologists and breast cancer surgeons to work together to deliver a full course of radiation treatment in one day to select women diagnosed with early-stage breast cancer. Traditional external beam radiation therapy (EBRT) involves daily radiation treatments for six to eight weeks, while IORT with the Xoft System can be completed in as little as eight minutes. IORT delivers a full course of targeted radiation from inside the body, directly within the tumor cavity, targeting cancer cells and reducing the risk of damage to nearby healthy tissue including tissue in the heart, lungs and ribs. A growing body of favorable clinical data supports the use of IORT in candidates meeting specific selection criteria.
“IORT with the Xoft System offers patients a number of unique benefits, including shorter treatment times, fewer side effects, reduced costs and added convenience compared to traditional treatment,” said Barbara Schwartzberg, M.D., FACS, breast surgeon, Sarah Cannon/HealthONE, Rose Medical Center, Denver. “By treating early-stage breast cancer in just one day, eligible patients are often able to return to their daily lives within days and better maintain their quality of life.”
The multidisciplinary Miami Breast Cancer Conference brings together surgical, medical and radiation oncology experts with the aim of fostering awareness of state-of-the-art treatments for breast cancer in each therapeutic area, encouraging cross-team cooperation in the clinic.
The Xoft System is U.S. Food and Drug Administration (FDA)-cleared for the treatment of cancer anywhere in the body, including early-stage breast cancer, non-melanoma skin cancer and gynecological cancers.
For more information: www.xoftinc.com


 May 07, 2026
May 07, 2026 









