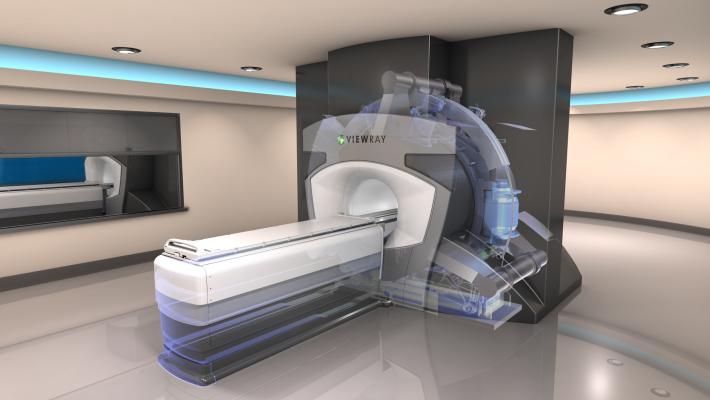
August 17, 2015 — ViewRay Inc. announced in late July its successful completion of a reverse merger with ViewRay Technologies Inc. The combined entity will focus solely on the business of ViewRay, the makers of MRIdian, the world's first and only commercial magnetic resonance image (MRI)-guided radiation therapy system that images and treats cancer patients simultaneously. ViewRay will trade on the OTC Markets under the symbol "VRAY."
ViewRay also announced that it has consummated a private placement with gross proceeds of approximately $26.7 million for the issuance and sale of 5,340,704 shares of its common stock. Proceeds from the private placement will be used to support the ongoing commercialization of MRIdian for research and development related to continued product development activities, and for general corporate purposes, including working capital. Investors in the private placement included Aisling Capital, Fidelity, Kearny Venture Partners, Montrose Capital, OrbiMed and Xeraya Capital.
Combined with other recent transactions, the company has achieved an approximate total of $76.7 million in financing, according to ViewRay CEO Chris Raanes.
In connection with the private placement, ViewRay has agreed, subject to certain terms and conditions, to file a registration statement under the Securities Act of 1933, as amended, covering the resale of the shares of common stock issued in the reverse merger and the private placement and in connection with the exercise of warrants issued to the placement agents, within 90 days after the closing. The shares of common stock issued or to be issued pursuant to the merger, the private placement and the placement agent warrants have not been registered under the Securities Act of 1933, as amended, or state securities laws and may not be offered or sold in the United States absent registration with the Securities and Exchange Commission or an applicable exemption from the registration requirements.
For more information: www.viewray.com


 May 06, 2026
May 06, 2026 









