September 19, 2011 — Trig Medical Inc. announced its participation at the International Society of Ultrasound in Obstetrics and Gynecology’s (ISUOG) 21st World Congress on Ultrasound in Obstetrics and Gynecology, September 18-22 at the Hyatt Regency Century Plaza in Los Angeles. Trig Medical is a health technology company specializing in labor progression management and interventional ultrasound guidance.
In addition to exhibiting, the company will have two short oral presentations (OP) and two electronic posters for review. All of their efforts will highlight a new standard in labor progression management with its new product LaborPro.
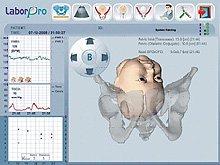
The system was launched in the United States at the American Congress of Obstetricians and Gynecologists (ACOG) annual clinical meeting in May 2011. It unites ultrasound imaging with position tracking to provide objective information and three-dimensional representations necessary to accurately assess labor progression and improve maternal and fetal care.
In addition, by reducing the need for manual exams during labor, the system improves patient comfort and mitigates the risk of infection to both mother and baby.
Two OPs highlighting LaborPro will be presented in the “Labor at Term” OP22 category Sept. 20 from 12:20 p.m. to 1:05 p.m. in the Olympic One room. In the first, Emma Bertucci, M.D., will discuss pelvimetry by the LaborPro system for the diagnosis of dystocia, or difficult or abnormal labor and delivery. Bertucci is a physician with the prenatal medicine unit in the department of obstetrics and gynaecology at the University of Modena in Modena, Italy.
Shoshana Haberman, M.D., director of perinatal services at Maimonides Medical Center in New York will present the second OP. She will speak on the evaluation of the occipito posterior position, or when the baby’s head is face up towards the mother’s abdomen, before head engagement; she will also discuss the risk of persistent occipito posterior and cesarean section.
In addition to the short OPs, two electronic posters (P) on LaborPro will be available for viewing Sunday, Sept. 18 to Thursday, Sept. 22 at the e-poster viewing stations in the California Lounge.
E-poster P16.05, authored by Bertucci, Vincenzo Mazza, M.D., and Carlotta Cani, M.D., et al., focuses on an innovative method of combining ultrasound and position tracking for three-dimensional continuous visual monitoring of vacuum extraction.
The second e-poster, P16.15, authored by Haberman, Yoav Paltieli, M.D., and P. Lynn, M.D., et al., documents mothers’ satisfaction with ultrasound based three-dimensional imaging of labor progression with LaborPro.
LaborPro is expanding its presence in United States healthcare facilities; the device currently has installations at Maimonides Medical Center in New York City, Hutzel Women’s Hospital in Detroit and the Washington Hospital Center in Washington, D.C.
For more information: www.trigmed.com


 May 07, 2026
May 07, 2026 









