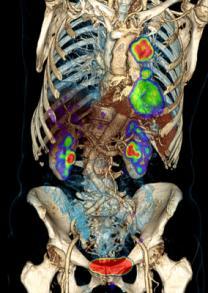
October 13, 2009 – Siemens Healthcare reported today at the annual meeting of the European Association of Nuclear Medicine (EANM) in Barcelona, Spain, that leading hospitals in England, France, Germany and Monaco have shown strong interest and adoption of the Biograph mCT, the worlds first molecular CT, to fuel advanced diagnostic capabilities and to drive greater workflow efficiencies.
The Biograph mCT combines the abilities of a high-resolution positron pmission tomography (PET) system with those of a premium diagnostic computed tomography (CT) system. This unique combination of technologies results in improved image quality reduced scan times and increased patient comfort.
First introduced at EANM in 2008, the Biograph mCT has a uniform 2-mm image resolution throughout the entire field of view designed to significantly improve image quality and offer clinicians increased confidence in diagnosis. The ability for five-minute, whole-body PET scanning, combined with a large bore and short tunnel, offers a more flexible solution for demanding workflows, as well as a more comfortable environment for the patient.
The Biograph mCT can serve as a PET/CT system for molecular imaging, and at the same time, it is powerful enough to serve as a dedicated CT. Among the first to work with the new system is Dr. Andreas Bockisch, director of the Clinic for Nuclear Medicine, University Clinic in Essen, Germany, and president of the German Society for Nuclear Medicine.
"Technology and the work environment are important factors in medical imaging diagnostics,” said Bockisch. “Improvements in image quality immediately increase diagnostic certainty.”
With the Biograph mCT, one examination room serves two modalities. PET and CT scans alike can be performed by one team of operators using only one schedule and. The system provides functional, anatomical, and molecular information from non-invasive examinations, plus a high level of efficiency. The High Definition PET (HD/PET) technology of the new system enables faster scan times, as well as sharper and more precise images. As compared to previous solutions, Biograph mCT is currently the only system that performs routine PET/CT scanning in only five minutes, and coupled with a homogeneous two millimeter resolution across the entire field of view, it produces high quality diagnostic images that surpasses standard PET images.
The compact design of the Biograph mCT also makes the system more patient-friendly than conventional systems. An oversized 78-cm bore and short tunnel, in combination with an extra-wide and comfortable patient table designed for a capacity of 227 kilograms, are highly suitable for patients previously considered difficult to examine, such as bariatric or geriatric patients.
Dr. Gerald Antoch, head of the department and assistant director at the Institute for Diagnostic and Interventional Radiology and Neuroradiology at the University Clinic Essen, envisions the combination of PET and high-performance Spiral CT opening up completely new possibilities in the area of hybrid oncological and cardiac diagnostics .
”It allows us to apply examination protocols specific to a particular disease in a patient-oriented manner,” said Antoch. “Some excellent examples of this are PET/CT colonographies for patients suffering from colon cancer or hybrid cardiac imaging for patients with coronary cardiac pathologies."
In addition, the TrueV option expands the axial field of view of the PET portion of Biograph mCT by one-third – increasing the performance of the system enormously. When required, the physician is able to individually reduce the dose or scan times, or considerably improve image quality.
The system is also equipped with a premium-class CT system that can adjust to a patient’s needs and medical requirements. As the first molecular CT scanner, Biograph mCT combines dynamic components, such as movable adaptive dose screening and a scan range of up to two meters. The scanner’s high temporal resolution of up to 74 milliseconds, combined with up to 128 slices per rotation, enables display of even the finest anatomical details at reduced motion artifacts. Using the Adaptive Dose Shield, the system avoids unnecessary radiation during all examinations. This ensures that the patient is exposed only to the clinically required dose.
In addition, the Biograph mCT has experienced widespread adoption because it also offers a shared-service model, where the same capital investment can serve both radiology and molecular imaging needs. From a bottom-line perspective, the use of one integrated scanner that offers shared services between molecular imaging and radiology departments is a highly efficient means of making the most of an institution’s imaging equipment and strengthening the existing workflows between the two departments.
For more information: www.siemens.com/healthcare


 May 19, 2026
May 19, 2026 









