September 29, 2015 — Seno Medical Instruments Inc. (Seno) announced it has completed the final phase of the company's U.S.-based PIONEER pivotal study of the Imagio breast imaging system. The system utilizes optoacoustic technology as a new tool to improve the process of diagnosing breast cancer.
The PIONEER Study was designed to demonstrate that the Imagio breast imaging system can provide physicians with vital information they need to determine whether a suspicious breast mass is cancerous or not, helping women avoid negative biopsy procedures. Results from this study will serve as the basis for the company's Premarket Approval Application (PMA) with the U.S. Food and Drug Administration (FDA).
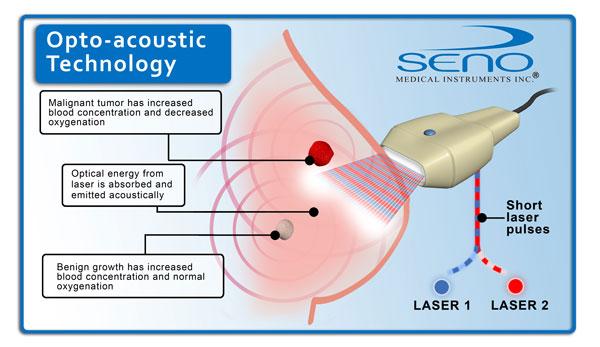
The system fuses optoacoustics, a technology based on "light-in and sound-out," with diagnostic ultrasound. The optoacoustic images provide a unique blood map in and around suspicious breast masses. Cancerous tumors grow relatively quickly and require significant amounts of blood and oxygen, so a network of blood vessels grows around cancerous masses. Imagio provides real-time images of these networks and a map of relative oxygen-rich or oxygen-deprived blood. Unlike other functional fusion technologies, Imagio uses no X-rays (ionizing radiation) or injectable contrast agents or radioisotopes to obtain its information, thereby reducing the patient's exposure to any potentially harmful aspects of imaging.
"In light of the fact that a large majority of biopsies reveal benign pathology, we believe that Imagio could potentially help reduce the number of procedures generated by false positives by providing physicians with more information and thus be more confident in their imaging assessments," said Erin Neuschler, M.D., Northwestern Medicine radiologist, assistant professor of radiology at the Northwestern University Feinberg School of Medicine and the co-principal investigator of the PIONEER Study. "Data from prior Seno studies demonstrated Imagio's potential to achieve clinically meaningful diagnostic sensitivity and specificity for breast cancer beyond those delivered by traditional, diagnostic ultrasound. It is our hope and belief that the results of this study will confirm these earlier findings."
"We started doing basic research with optoacoustics back in 2005 and it is noteworthy to see this rigorously conducted trial of this technology completed. We await the final analysis now that all of the patients have enrolled in the trial," said Stephen Grobmyer, M.D., director of breast services and principal investigator of the trial at Cleveland Clinic. "Based on our clinical experience to date, it appears this breast cancer imaging technology could enhance both the physician and the patient experience by reducing the number of false positive cases, which we currently face assessing patients with abnormal breast imaging."
The study was conducted in 16 leading institutions throughout the United States, with more than 2,100 subjects enrolled. The study was designed to measure the sensitivity and specificity of Imagio compared to grayscale ultrasound imaging in breast lesions using the probability of malignancy (POM). Subjects underwent a traditional ultrasound and an Imagio scan. The radiologist used clinical mammography and traditional ultrasound findings to determine if the subject should advance to the biopsy phase. The Imagio results were later interpreted by an independent reader panel. Subjects who were put into follow-up were re-evaluated 12 months after their initial examination to confirm the results as a true negative.
After the interim analysis of the PIONEER Study, Seno initiated a separate European post-marketing and clinical follow-up study, the MAESTRO Study. The company currently has five sites participating in this study in the Netherlands.
Results from the MAESTRO Study will be presented at the upcoming 2015 European Society of Breast Imaging Annual Scientific Meeting (EUSOBI), Oct. 2–3 in London. The results of the PIONEER Pilot Study will be presented at the Radiological Society of North America (RSNA) annual meeting, Nov. 29 – Dec. 4 in Chicago.
Seno Medical Instruments, Inc. has provided support for the research done at Cleveland Clinic and Northwestern University and paid for travel related to the participation in the trial for Grobmyer and Neuschler, respectively.
For more information: www.senomedical.com


 May 07, 2026
May 07, 2026 









