March 22, 2023 — The Riverain Technologies ClearRead Bone Suppression solution is now available on the Siemens Healthineers syngo.via OpenApps, bringing revolutionary Clear Visual Intelligence (CVI) to radiologists in Europe and the U.S. using the syngo.via diagnostic imaging platform.
ClearRead is the only proven CVI solution with suppression technology that enables radiologists to go beyond the standard background-impaired imaging interpretation to see past obstructions like bones and vessels to correctly and quickly detect cardiothoracic diseases with newfound Certainty of Search (CoS). ClearRead leverages artificial intelligence and deep learning to create advanced modeling that improves lesion conspicuity and measurement precision.
“Lung cancer is the number one cancer killer in Europe and the U.S., but when lung cancer is found early, survival rates are dramatically higher,” says Steve Worrell, CEO of Riverain Technologies. “The extension of our long-time partnership with Siemens that supports an enterprise solution is an exciting extension – allowing a standard of care and potential to detect lung cancer incidentally sooner with a single X-ray exposure without any additional effort for technologists.”
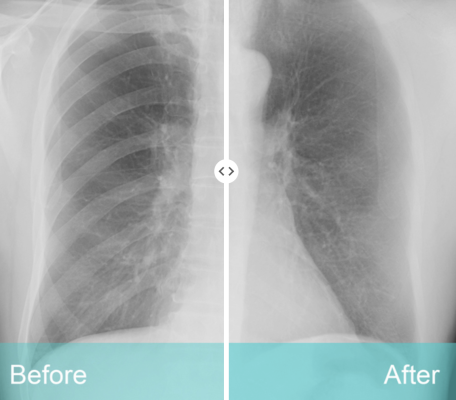
While traditional X-rays help identify lung abnormalities, the body’s 12 sets of ribs and clavicles stand in the way of a clear view. Radiologists are often at the mercy of what their eyes can see. ClearRead’s unique suppression technology removes obstructions from an image, allowing the radiologist to detect actionable nodules more effectively on frontal chest X-rays.
ClearRead X-ray bone suppression helps radiologists detect 1 out of 6 previously missed nodules on an image (17% improved nodule detection)1, and it significantly improves fungal infection detection2.
Siemens Healthineers syngo.via OpenApps allows syngo.via RT Image Suite customers to purchase the ClearRead X-ray solution from the Siemens Healthineers Digital Marketplace, which connects customers to a portfolio of applications from partners like Riverain. It enables Siemens Healthineers European and U.S. customers to browse, test, and download apps, as well as request quotations.
ClearRead X-ray is device and protocol agnostic and works with all manufacturers’ X-ray devices. It does not require changes to existing imaging workflows, and it does not require an additional radiation dose.
For more information: https://www.riveraintech.com/


 May 27, 2026
May 27, 2026 









