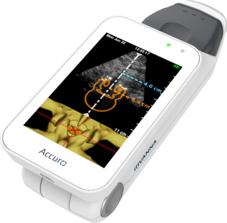
Accuro image courtesy of Rivanna Medical
July 14, 2015 - Rivanna Medical announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance to market Accuro, a handheld and untethered smart phone-sized ultrasound device. Accuro is designed to guide spinal anesthesia with automated 3-D navigation technology in addition to ultrasound imaging of abdominal, musculoskeletal, cardiac and peripheral vascular anatomies.
The platform is designed to provide automated navigation to an anatomical target so a clinician may avoid "guessing" where the target is. The initial commercial application is designed for guiding clinicians to a first-attempt success in administering spinal anesthesia.
Rivanna plans to launch Accuro at the annual scientific meeting of the American Society of Anesthesiologists (ASA) in San Diego in October 2015.
For more information: www.rivannamedical.com


 May 05, 2026
May 05, 2026 









