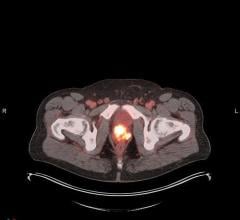
September 23, 2009 - The results of the Phase 0 (exploratory), or “first-in-human,” study indicated that the bio-distribution of the new agent was recorded at safe levels for clinical use in positron emission tomography (PET) studies, according to a presentation at the World Molecular Imaging Conference in Montreal, Canada.
Siemens Healthcare announced today the initial findings of a new imaging biomarker developed by its molecular imaging biomarker research facility in collaboration with Dr. Jian Q. (Michael) Yu and Fox Chase Cancer Center in Philadelphia, Penn. "These are very exciting initial results," said Jian Q. Yu, M.D., of Fox Chase Cancer Center. "The potential ability to non-invasively determine the level of potency in malignant cancer cells might drastically change the treatment course in individual patients."
The new imaging agent was designed to capture and quantify the cellular expression of CA-IX, an enzyme whose expression has been linked to tumor growth and invasion, as well as hypoxia. CA-IX is an active enzyme, whose expression usually promotes tumor growth and invasion, and thus impairs prognosis. Noninvasive quantification of CA-IX expression may have the potential to characterize the biologic aggressiveness of malignant tumors in an individual, without the need for the invasive collection of tissue samples. The detection of CA-IX in patients may provide a novel approach to predict disease outcomes and efficacious responses to cancer therapy, and, ultimately, help physicians decide on the most effective treatment options for patients.
The agent was also found to be stable for imaging at 133-minutes post injection, which allows a sufficient window for acquiring the image, and that the agent safely clears the body through urinary elimination. The study included initial human data regarding bio-distribution of the new agent, radiation dosimetry levels in normal volunteers and optimal patient imaging parameters using PET. Further study with patients is currently in progress.
Carbonic anhydrase IX (CA-IX) is one subtype of transmembrane carbonic anhydrases, which catalyzes the reversible hydration of carbon dioxide into carbonic acid and is believed to be involved in pH regulation, which is linked to tissue invasion by tumors. The absence of CA-IX expression has been observed in many normal tissues. The abnormal expression of CA-IX has been detected in many carcinomas originating from CA-IX negative tissues including, the brain, kidney, lung, breast and uterine cervical tissues.
Phase 0 trials are conducted in accordance with the Food and Drug Adminstration’s (FDA) guidance on exploratory investigational new drug (IND) studies. These types of studies are intended to improve the development of promising drugs or imaging agents by determining at a very early stage if the drug or agent performs as expected, based on earlier results in pre-clinical trials.
For more information: www.siemens.com/healthcare and www.fccc.edu


 May 29, 2026
May 29, 2026