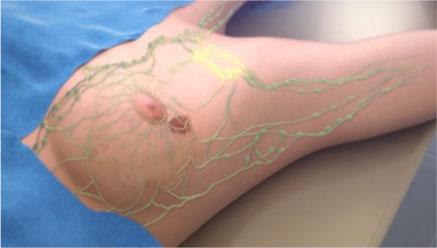
Image courtesy of Navidea Biopharmaceuticals Inc.
September 3, 2015 — Navidea Biopharmaceuticals Inc. received initial notice of award for a Fast Track Small Business Innovation Research (SBIR) grant up to $1.8 million from the National Cancer Institute (NCI), National Institutes of Health (NIH). The grant will be used to fund preclinical studies examining the safety of intravenous (IV) injection of Tc99m tilmanocept, a Manocept platform product. This will be followed by a clinical study of the safety and efficacy of single photon emission computed tomography (SPECT) imaging studies to identify and quantify both skin- and organ-associated Kaposi’s Sarcoma (KS) lesions in human patients.
The SBIR grant is awarded in two parts, with the potential for total grant money of up to $1.8 million over two-and-a-half years. The first six-month funding segment of $300,000, which has already been awarded, is expected to enable Navidea to secure necessary collaborations and Institutional Review Board (IRB) approvals. The second funding segment could provide for up to an additional $1.5 million to accrue participants, perform the Phase 1/2 study, and perform data analyses to confirm the safety and effectiveness of intravenously administered Tc99m tilmanocept.
“Building on earlier research showing KS tumor cells abundantly express CD206, the tilmanocept receptor, and successful imaging with subcutaneous injection, we plan to evaluate intravenous administration of this targeted imaging agent to noninvasively locate internal KS lesions, which are currently challenging to identify and monitor,” commented Toby Maurer, M.D., FAAD, professor of dermatology at the University of California, San Francisco (UCSF), and chief of dermatology at San Francisco General Hospital and Trauma Center, who is co-PI at UCSF with Michael S. McGrath, M.D., PhD. “If successful, future diagnostic and eventually therapeutic developments have the potential to dramatically improve life expectancies and quality of life for patients suffering from Kaposi’s sarcoma.”
For more information: www.navidea.com


 June 01, 2026
June 01, 2026 









