
ExactVu micro-ultrasound system image courtesy of Exact Imaging.
July 28, 2016 — Exact Imaging announced that The Journal of Urology has published the initial results supporting the development of their PRI-MUS (prostate risk identification using micro-ultrasound) protocol. The study is published in the August 2016 issue of the American Urological Association’s official journal, and the peer-reviewed paper is now available online.
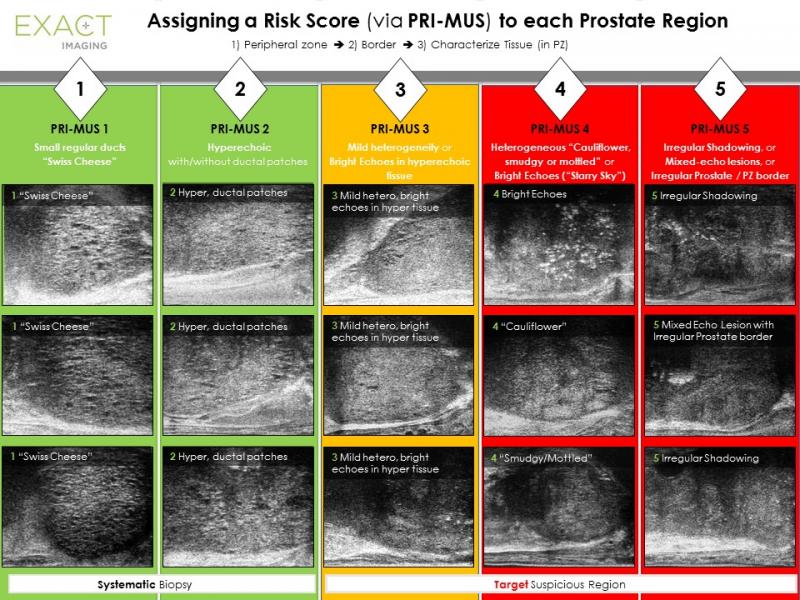
The paper illustrates the initial finding on PRI-MUS, the first risk identification protocol developed specifically for micro-ultrasound of the prostate. This protocol aims to set a new standard to facilitate micro-ultrasound-based visualization and stratification of prostate tissue imaging and to best instruct real-time targeting of suspicious regions using the micro-ultrasound platform.
"This paper reveals the promising initial results for real-time visualization of suspicious lesions and targeting of biopsies using micro-ultrasound, along with the PRI-MUS protocol," said Sangeet Ghai, M.D., PRI-MUS Committee chair and director of the Biopsy Centre at Princess Margaret Cancer Centre, Toronto, Ontario. "As we concluded in the paper, the improved performance of this new imaging platform and the protocol in detecting significant disease is consistent with the focus of the field on increasing detection of high-risk disease early and on decreasing insignificant diagnoses."
The goal, according to Exact Imaging President and CEO Randy AuCoin, was to develop an evidence-based protocol that would allow urologists to identify sonographic features now visible in prostate tissue, characterize those features in terms of a standardized risk identification protocol and target those areas that appeared suspicious. AuCoin said the protocol was developed based on retrospective studies referencing over 1,000 micro-ultrasound biopsy images and cine-loops, all validated with clinical pathology.
Along with Ghai, the other members of the board who helped develop and validate the protocol are: Christian Pavlovich, M.D., Johns Hopkins University, Brady Urological Institute, Baltimore; Gregg R. Eure, M.D., Urology of Virginia, Virginia Beach, Va.; Vincent Fradet, M.D., CHU de Quebec - Université Laval - L'Hôtel-Dieu de Québec, Quebec, QC; and Eric Hyndman, M.D., Ph.D., Prostate Cancer Calgary, Calgary, Alberta.
Exact Imaging is planning on CE approval and then U.S. Food and Drug Administration (FDA) clearance for the ExactVu micro-ultrasound system in the coming months. The company's Quality Management System has just been assessed and recommended for ISO 13485:2003 certification, a key precursor to CE approval and 510(k) clearance.
For more information: www.exactimaging.com


 April 09, 2026
April 09, 2026 









