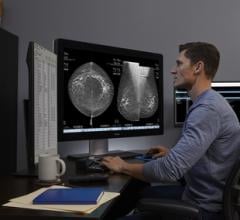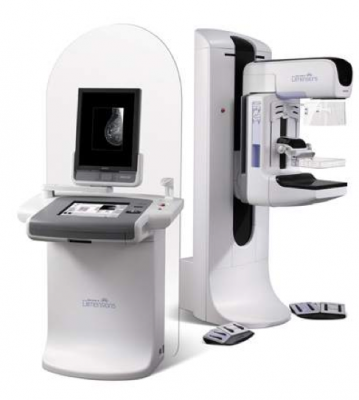
At RSNA 2011, Hologic will feature its Selenia Dimensions 3-D breast tomosynthesis technology. It is designed to help clinicians in catching breast cancers earlier and in significantly reducing the number of unnecessary diagnostic work-ups.
Launched in internationally markets in 2008, Selenia Dimensions breast tomosynthesis was approved for sale in the United States in 2011. Selenia Dimensions breast tomosynthesis delivers the exceptional 2-D images and, for the first time ever, allows a radiologist to offer their patients a conventional 2-D digital mammogram and a 3-D tomosynthesis exam in one compression in seconds. The combination of measurable improvements in accuracy and detection, and improved sensitivity, makes the new system a superior system vs. conventional digital mammography systems.
Unlike prior-generation mammography systems which generate two-dimensional images, a breast tomosynthesis system produces three-dimensional images which are intended to reveal the inner architecture of the breast, free from the distortion typically caused by tissue shadowing or density.
Hologic is offering five 90 minute programs combining lectures and hands-on experience reading 3-D breast tomosynthesis cases. The programs are designed for radiologists, administrators, technologists and others interested in learning more about this recently approved breast cancer screening and diagnosis technology.
More than a dozen scientific papers and poster session by clinicians using tomosynthesis in their practice will be presented at RSNA. Consult the RSNA Meeting Program Guide for times and places.
For more information: www.hologic.com


 April 20, 2026
April 20, 2026