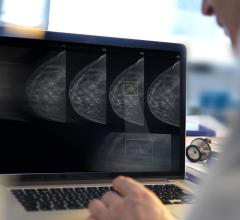
Aug. 4, 2009 - The FDA gave Cerner 510(k) pre-market clearance for the ProVision Workstation – Mammography solution, which is designed to help radiologists efficiently interpret the growing number of mammography images.
According to the American Cancer Society, the number of women age 40 and above who have received a mammogram in the past two years has grown from 29 percent in 1987 to 70 percent in the year 2000.
When radiologists review images, they typically use separate viewers to study images like X-ray or CT scans and mammography images. The ProVision Workstation – Mammography solution eliminates the need for separate general imaging and mammography viewers, reducing the inefficiencies and delays created when using different viewing systems. Radiologists can view all imaging modalities, including MR, PT, US, CT, XR, XA and MG, and the patient’s electronic health record (EHR) from one workstation. By breaking the traditional model of dedicated mammography and general radiology workstations, Cerner is maximizing radiologists’ efficiency and increasing the imaging department throughput.
For more information: www.cerner.com


 July 30, 2025
July 30, 2025