May 13, 2019 — Zebra Medical Vision has received U.S. Food and Drug Administration (FDA) 510(k) clearance for HealthPNX, an artificial intelligence (AI) alert for pneumothorax (PNX) based on chest X-rays.
The latest FDA clearance, received in May 2019, focuses on an AI alert for "stat" (urgent) findings of pneumothorax, and demonstrates a promising potential to substantially reduce turnaround time and increase the radiologist's confidence in making this diagnosis. Chest X-rays are one of the world's most used imaging modalities.
Pneumothorax is a condition in which there is an accumulation of gas within the pleural space between the lung and the chest wall. Without prompt management, pneumothorax can lead to total lung collapse and other potentially fatal complications. This condition is most commonly diagnosed by a chest X-ray scan, though it is one of the hardest to interpret, and is known for high disagreement rates even between experienced radiologists. Misdiagnosis or late-diagnosis of Pneumothorax impacts around 74,000 Americans per year.1
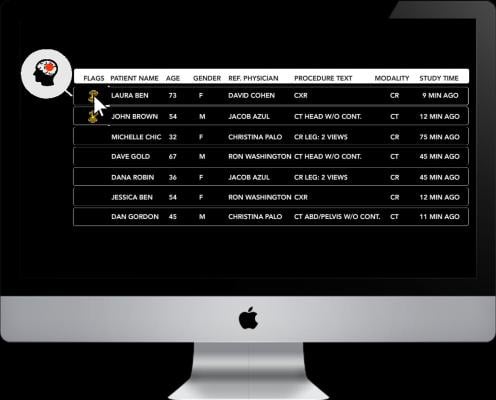
The patent pending technology automatically detects findings suggestive of pneumothorax based on CXR or digital radiography (DR) scans, and alerts the medical team. In hospitals where Zebra-Med's "All in one" (AI1) solution is integrated into the radiologist's worklist, the scan is flagged so that the radiologist can address it in a timely manner. This first-of-its-kind FDA-cleared solution can save physicians more than 80 percent of the time taken to reach the acute condition, compared to the traditional First In First Out (FIFO) methodology, according to Zebra Medical.
The chest X-ray AI network was trained using millions of images to identify more than 40 common clinical findings. The results of the Textray study demonstrate high rates of agreement between the algorithm and human radiologist experts.2
"In a clinical validation study we performed, Zebra-Med's acute CXR pneumothorax and CT Brain bleed products demonstrated a promising potential to substantially reduce turnaround time and increase the radiologist's confidence in making these diagnoses," said Terence Matalon, M.D., chairman of imaging at Albert Einstein Medical Center. Zebra-Med's AI1 Triage Solution is the first of its kind for both computed tomography (CT) scans and X-rays, and currently addresses two acute conditions: intracranial hemorrhages (head CTs, FDA-pending), and pneumothorax (chest X-rays).
For more information: www.zebra-med.com
References
1. Bintcliffe O., Maskell N. Spontaneous pneumothorax. British Medical Journal, May 8, 2014. doi: https://doi.org/10.1136/bmj.g2928


 May 12, 2026
May 12, 2026 









