The Trilogy console.
The advent of conformal radiotherapies has ushered in an era of image-guided radiotherapy, or IGRT, enabling clinicians to compensate for the geometric uncertainties inherent in the radiotherapy (RT) delivery process. A diverse set of imaging tools has been added to the radiotherapy treatment vault, making it possible to deal with inter-fraction motion (i.e., changes in target position due to set-up error or naturally-occurring changes in organ position over time) and intra-fraction motion (i.e., organ motion during treatment, usually due to respiration or other physiological processes).
Key Imaging Tools
Three important imaging tools are included in most IGRT technology packages from Varian Medical Systems. These are: the PortalVision MV imaging system; the On-Board Imager–kV imaging system for generating high-resolution radiographic (kV), fluoroscopic, and cone-beam CT images right at the treatment machine; and the Real-Time Position Management™ (RPM) system for managing motion during imaging and treatment.
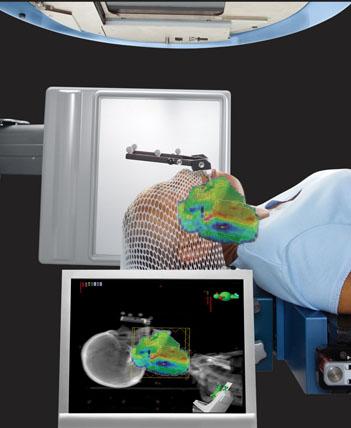
Outfitted with these image-guidance tools, a Varian linear accelerator offers many choices for treating different disease types. Doctors can use orthogonal pairs of kV and/or MV radiographic images for patient repositioning based on bony anatomy or fiducial markers; volumetric cone-beam CT imaging for patient repositioning based on soft tissue structures, and fluoroscopic imaging for gating verification. For the most part, images are acquired just after the patient has been positioned for treatment, and then special matching software is used to compare the images to reference images from the treatment plan. The system then calculates a “shift” and sends instructions to the treatment couch, which moves to bring the targeted tumor into precise alignment for treatment.
For clinicians in departments that are deploying these technologies for the first time, the prospect of setting up an IGRT program can seem daunting. It requires establishment of protocols that specify which tools to use, and when. And everyone—from the doctors and physicists to the dosimetrists and therapists—must acquire new skills and work together in unfamiliar ways.
This article summarizes the experience of two early adopters who were instrumental in setting up IGRT programs at their respective treatment centers. The centers are about as different as two cancer treatment facilities can be. One is a small, single physician practice; the other is a large, multi-site, research oriented institution in a major metropolitan area.
Image Guided Radiation Therapy at Community Care Physicians in Latham, N.Y.
Image Guided Radiation Therapy is a small, single-physician radiation oncology practice that was also one of the earliest adopters, in mid-2005, of Varian’s On-Board Imager system for image-guidance. Approximately 95 percent of the patients receive IMRT, primarily for the treatment of prostate cancer. All IMRT treatments are image-guided, either using kV-kV matching or cone-beam CT.
Robert Desjardins, RTT, MBA, the site’s administrative director, identifies a few “success factors” that help with the establishment of an IGRT program. They are:
1) having a project champion, preferably the
radiation oncologist;
2) working with the clinical team to help them acquire the skills for identifying anatomical structures in CT data sets, and;
3) involving IT personnel from the beginning, so that storage-intensive information systems are properly configured to support departmental
workflow.
“Adopting new technology requires change. A local champion can foster buy-in so the entire team can feel more comfortable, and I think it’s best if that person is the physician. Once that leader is in place, you can assign a project leader to reach out to the vendor, help set up the time line for technology installation and acceptance, and start working with IT personnel, because IGRT is IT-intensive. After that, it’s about training and practice.”
According to Desjardins, positioning patients using the kV-kV image matching procedure was relatively easy for therapists to learn. “It’s like something they are already used to seeing; it’s like a port film, but the resolution is much higher. The cone-beam CT images are the best when it comes to image guidance because you can see everything—soft tissue, bony anatomy, everything, and in three dimensions. So therapists are less familiar with these, and there’s a slightly steeper learning curve involved. I’d say any therapist can learn to read and act on the CT images in less than two weeks.”
The majority of patients receive 7-field IMRT treatments for prostate cancer. Once therapists are fully trained and comfortable with the IGRT process, the total amount of time needed to complete the process is about 12 minutes. The cone-beam CT image takes three minutes (including a 30 second “mode up,” a one-minute acquisition, and five seconds for image reconstruction). Image matching and executing couch shifts adds another
1-2 minutes.
The protocol calls for a physician consultation if shifts are greater than half a centimeter in the lateral direction, or greater than one centimeter in the superior–inferior or anterior–posterior directions. So long as the shifts are within tolerance, the
therapist can proceed with the treatment. If something is outside the tolerance, they call the physician.
Henry Ford Health System, Detroit, Mich., and Surrounding Communities
According to Teamour (Tim) Nurushev, Ph.D., director of clinical physics for the Henry Ford Health System (HFHS), dosimetric consequences should be central among the considerations that go into designing an IGRT protocol. For example, he stated, you typically would not perform a daily cone-beam CT match for a conventionally-fractionated lung cancer treatment, although you would for a radiosurgery (five fractions or less). “In the case of the radiosurgery, the benefit of lining up the patient correctly outweighs the potential risk of delivering the extra dose. For a normal lung treatment, when you’re delivering multiple fractions, using cone-beam CT every day could end up delivering more dose than the lung tissue can tolerate. The point is: IGRT protocols must be driven by physics and dosimetry, and so we create the protocols in tight consultation with the physicians. We work together to find the most suitable way to set a patient up while still maintaining minimal dose to the patient.”
In addition, imaging protocols should also help clinicians to feel comfortable with what they’re doing, Nurushev says. For example: most radiation oncologists have gotten used to seeing MV images when treating breast cancer, and they want to see how the field aperture correlates to the patient’s anatomy, to understand how much lung or skin may be in the treatment field. This is only visible with MV imaging.
At HFHS, physicians and physicists have collaborated in developing the IGRT protocols that are now in use. Most are a variation of one of three approaches: 1) daily cone-beam CT imaging; 2) daily kV-kV imaging with a weekly cone-beam CT, and 3) more complex SRS/SBRT protocols:
1) Head and neck treatments involve kV-kV matching to some fixed piece of the skull or jaw in close proximity to the tumor. “We don’t do a daily cone-beam CT because we are comfortable that we’re getting two millimeter precision based on kV matching to bony anatomy,” Nurushev said. “However, patients often respond well to treatment, and the tumor often shrinks significantly, so we do a cone-beam CT once a week. The physician evaluates the images and decides when to replan. We are now investigating the possibility of an approach that would involve replanning using the cone-beam CT images, rather than doing another CT simulation.”
2) Prostate cancer patients receive a daily cone-beam CT, which adds the equivalent of one fraction’s worth of dose to the 42 fractions delivered. “We deliver high doses, between 78-80 Gy, so we feel the cone-beam CT is essential for accurate alignment every day,” Nurushev said.
3) For conventionally-fractionated lung treatments, planning is done using 4D CT and PET data, and patients are set up for daily treatments using kV-kV matching. For SBRT, set ups are done using cone-beam CT. “We also use weekly MV imaging to verify the ports for conventional 3D treatments,” Nurushev said. “It’s an added check for us.”
Clinical Training
One of the challenges in establishing an IGRT program at a large, multisite health system is clinician training. At HFHS, the physics and dosimetry teams trained the radiation therapists to read and evaluate images. “Many therapists were trained in film-based environments, and so were not used to looking at anatomy in three dimensions,” Nurushev said. “We spent the first few weeks with them, showing them how to do a manual image registration and compare that to an automatic one, to see which was better. We eventually dropped back to stand behind and support their efforts, until they could operate independently. They are empowered to stop a treatment if, after everything, they can’t get a good match.”
In addition, the physics staff has created detailed documentation in the form of brief, step-by-step guides with action criteria that specify the steps for different types of treatments. For example: therapists can go ahead and treat after making shifts up to one centimeter for breast cancer patients, but for prostate treatments, where the margins are only six millimeters, shifts larger than that must be checked by the physician.
Like Desjardins, Nurushev feels that the successful launch of an IGRT program requires internal “champions” to promote the project and overcome any aversion that the prospect of change might engender among busy clinicians. “You need the participation of your whole team to establish the most efficient protocols that do the best job of serving your patients,” he said.
“It’s a matter of getting motivated and making it happen,” Desjardins says. “That means
understanding the benefits to patients, and getting comfortable with making clinical judgments based on different kinds of image data. The rewards far outweigh the costs. IGRT can improve the quality of treatments. You’re able, for the first time, to see exactly what you’re treating, and based on our experience, you’ll never want to go back to treating without it. Members of our clinical staff now don’t feel comfortable delivering a treatment unless they use image guidance.”


 May 19, 2026
May 19, 2026