November 1, 2023 — The American Medical Association (AMA) has announced its 2024 Current Procedural Terminology (CPT) code set will include a new Category III code for Avenda Health’s innovative Unfold AI™, a cancer AI platform that creates new clinically meaningful insights to aid physicians and patients in making informed treatment decisions. CPT codes are used to report medical procedures and services for processing claims, conducting research, evaluating utilization and developing medical guidelines. The Category III designation is a temporary code for new and developing technologies.
The comprehensive description of the new code is:
X237T- Non-invasive prostate cancer estimation map, derived from augmentative analysis of image-guided fusion biopsy and pathology, including visualization of margin volume and location, with margin determination and physician interpretation and report.
“This recognition by the CPT Editorial Panel is a testament to our dedication to providing cutting-edge solutions in the field of cancer diagnosis and treatment,” said Dr. Brittany Berry-Pusey, COO and co-founder. “The new CPT code solidifies our position as a frontrunner in revolutionizing cancer care and our commitment to making our technology accessible to all patients.”
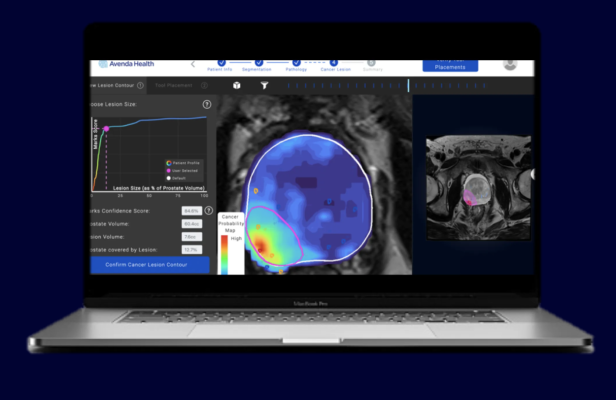
Unfold AI is a decision support platform that provides the physician and the patient with critical information, comprehensive insights, and planning guidance to ensure the patient is put on the right path for their unique cancer journey. By integrating patient-specific data from prostate imaging and biopsies the platform generates a customized cancer probability map using state-of-the-art machine learning. Its 3D, AI-generated map enables physicians to visualize the precise location of cancer for informed intervention planning.
Avenda Health recently announced Unfold AI’s use by Matthew Allaway, D.O. at Urology Associates in Maryland, leveraging the FDA-cleared technology in their procedures. Avenda plans to expand to major teaching hospitals and urology practices nationwide, ensuring widespread access to advanced cancer care solutions.
The full set of Category III codes are published annually in the CPT codebook. The most recently approved codes will be posted to the AMA CPT website January 1, 2024 with an effective implementation date of July 1, 2024.
For more information: avendahealth.com


 May 27, 2026
May 27, 2026 









