
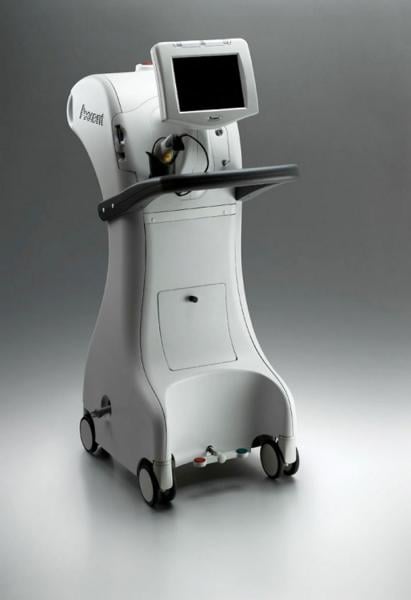
The Xoft Axxent system has a miniature HDR X-ray source and does not require extensive shielding of the radionuclide, allowing all personnel to remain in the OR.
The unfortunate reality of living with cancer is that oftentimes, the treatment can be just as much of a burden for patients as the disease itself. Once the tumor is removed, they must still undergo radiation therapy to try to prevent recurrence, and in some cases that means going back to the hospital every day for several weeks. This can be a taxing and expensive proposition for many patients, especially if they are traveling out of their immediate area for specialized care.
While these concerns are legitimate, the fact is that patients cannot receive the full benefit of radiation therapy if they do not receive the full course of treatment they are prescribed. For some of these patients, an answer has emerged in the last few years in intraoperative radiation therapy (IORT). While clinical evidence supporting its efficacy has been somewhat slow to emerge, many in the medical field are confident the technology is poised for a surge: The Cleveland Clinic included IORT at No. 9 on its annual Top 10 Medical Innovations list for 2015, a ranking assembled through voting by 156 physicians.
Clinical Benefits of IORT
The ability to drastically shorten the duration of radiation treatment may be the single greatest clinical benefit of IORT, and it could change the way patients view radiotherapy. A 2005 study in the Journal of Clinical Oncology analyzed how distance from the radiation treatment facility impacted the choice between mastectomy and lumpectomy, which requires follow-up radiotherapy, for 20,094 early-stage breast cancer patients. Forty-three percent elected mastectomy and 53 percent chose lumpectomy (the remaining 4 percent were unknown). Results indicated that decisions for mastectomy increased with distance from the radiotherapy facility.1
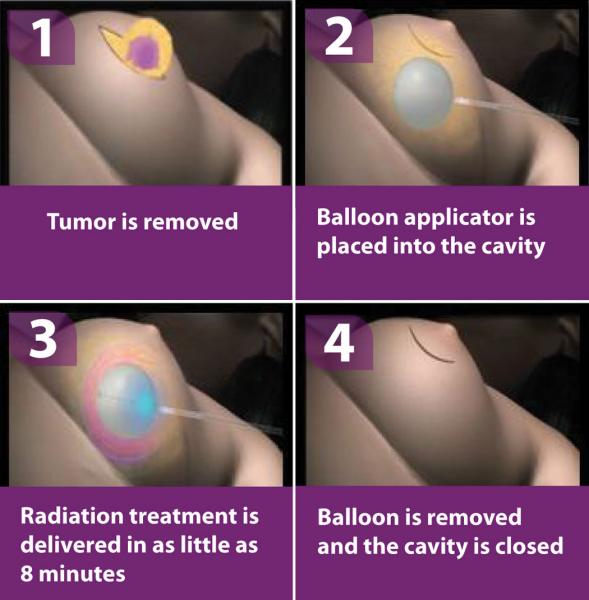
IORT delivers the full radiation dose during the surgery immediately after the tumor has been excised. The technique is a form of brachytherapy, with the dose targeted directly into the tumor bed, which is the most likely place for recurrence. The length of the procedure varies depending on the condition as well the IORT system being used, but in some cases it can be done in as little as 8 minutes. It can be delivered as its own full-course treatment, or as a radiation boost after that patient has already undergone several weeks of therapy.
Being able to start radiation therapy almost simultaneously with tumor removal dramatically cuts down on the amount of time required for radiation therapy. In breast cancer cases — IORT’s most common application — the entire course of treatment can be delivered in a single day, a variation on accelerated partial breast irradiation (APBI).
The direct dose application also minimizes accidental exposure to surrounding tissue and organs, including the skin — a distinct advantage over traditional external beam radiotherapy. “You get a higher radiation dose because you’re not going through those other structures,” said Jay Harness, M.D., FACS, breast surgeon at St. Joseph Hospital of Orange, Orange County, Calif. To further ensure the patient is protected, both internal and external shielding are used during IORT — internal protection usually consists of stainless steel plates inserted around the target area, while external drapes and other devices protect the patient as a whole, as well as the rest of the OR. The type of shielding required or included varies from system to system.
The Big Three
While IORT may just be coming into prominence now, the concept has been around for decades. One of the biggest problems, according to Rachael Bennett, RT(R)(T), clinical analyst for MD Buyline, was the reliance on traditional linear accelerators (linacs), which tend to be very large — and expensive. This puts hospitals in a difficult financial position.
“Some hospitals that can’t afford to have a dedicated room in their OR for radiation treatments would have to completely clear the schedule for the linac in the oncology department, make sure that everything in it was sterile, and maintain a sterile environment from the OR through the process of transporting a patient down to the linac,” she said. “That’s not very cost-efficient, not very safe for maintaining a sterile environment, and it’s very complicated and time-consuming.” Manufacturers have been working to create a solution to this dilemma, and that has resulted in smaller, more portable units from all three of the major vendors — Xoft, IntraOp and Zeiss.
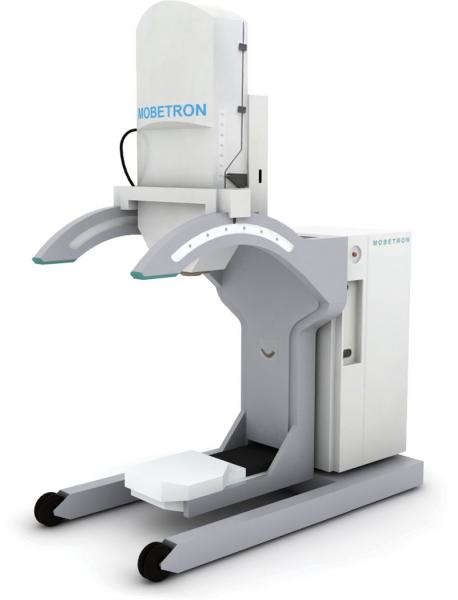
The essential difference between IORT systems is the type of radiation source. Electron IORT systems, like IntraOp’s Mobetron, employ a miniature linear accelerator to deliver dose to the patient. The use of electrons allows the dose to stay uniform across the entirety of the treatment area. Other systems, like the Xoft Axxent eBx and Zeiss’ Intrabeam, use miniature X-ray sources. Both units generate a low-energy
(50 kV) dose that is strongest at the applicator, but falls off rapidly as it progresses outward, helping limit dosage to nearby organs.
Shielding measures also vary from system to system. Mobetron, for example, is self-shielded, requiring no additional protection for patients or surgical staff. The Xoft Axxent system, with its miniature HDR X-ray source, does not require extensive shielding of the radionuclide, allowing all personnel to remain in the OR.
Applications for IORT
Though the benefits of IORT are well-known in many oncology circles, it has yet to achieve widespread adoption, largely due to a relative lack of clinical evidence supporting its effectiveness.
The technology has been most readily accepted in the treatment of early-stage breast cancer as a form of APBI, with several major clinical trials demonstrating its efficacy. In the United States, that position has largely been a product of the TARGIT-A trial, with results published in 2013. During the 12-year study, early-stage breast cancer patients were randomized to treatment with IORT or
whole-breast external beam radiation therapy (EBRT); the former was found to be non-inferior to EBRT. IORT patients did demonstrate a five-year
risk for local recurrence of 3.3 percent compared to 1.3 percent for EBRT. All told, approximately
80 percent of the IORT group completed their surgery and radiotherapy in one session without the need for any supplemental EBRT.2
“Breast cancer is really common, and because we’re detecting it at such an early stage these days, I think there’s a lot more women who are candidates for this type of surgery,” said Bennett. She noted that the TARGIT-B trial is already underway, expanding the focus to patients with a higher chance of recurrence. The study is targeted for completion in 2022.
As with other applications, the therapy can be administered as a full course or a boost. “APBI tends to be for low-risk women; boost can actually be more targeted toward the higher-risk patients or those who are more conservative and don’t want to do anything outside the standard of care,” said Derek DeScioli, vice president of global sales for IntraOp.
In addition to confirming the effectiveness of IORT, the TARGIT-A trial also helped identify criteria for patient selection. These criteria have been put into practice by users such as Olga Ivanov, M.D., FACS, medical director of the Breast Health Center at Celebration Health in Florida. Factors that Ivanov and her team select for include:
Histology of the tumor (patients with invasive ductal carcinomas are excluded);
Age — Patients over 40 years old (or 45, according to Ivanov) are best suited for IORT; and
Size of tumor — Tumors under 3 cm in size are not considered for IORT.
Ivanov estimates that even with these strict criteria, 40 percent of patients are eligible for IORT procedures, and 30 percent are considered “ideal” candidates.
Other Applications
Aside from its breast cancer applications, there are a few other cancers where IORT is being tested, but there is little clinical evidence to support many of these applications to date. Ken Ferry, CEO of iCAD, reports that the most popular application for the Xoft eBx system in the United States is actually in non-melanoma skin cancer, as clinicians are able to administer surface brachytherapy without the use of a radioactive isotope or a megavoltage linac. Outside the United States, the company has seen strong interest in IORT for endometrial and cervical cancer.
The Mobetron system has been utilized for a number of locally advanced and recurrent cancers, including pancreas, colorectal, sarcomas and head/neck cancers, according to DeScioli. “These tend to be cancers that are very difficult to operate on and be confident that you were able to get clear margins,” he said. There has also been limited exploration into IORT use for lung, prostate, gastrointestinal, pediatric and bladder cancers, according to DeScioli, but the number of patients is relatively low and data has been limited.
The Future of IORT
If IORT is truly going to secure a primary position in the radiation oncologist’s armamentarium, according to multiple sources, a more encouraging reimbursement scenario needs to be established. iCAD’s Ferry noted, “The Affordable Care Act requires hospitals to use the most effective care methodologies and technologies at the lowest cost,” suggesting that the reduced treatment times and lower system costs of IORT could drive it into those categories. The American Medical Association has released Current Procedural Terminology (CPT) codes related to IORT — two for treatment delivery (2012) and one for treatment management (2013).
For hospitals wanting to undertake IORT, current users say a team-based approach is critical to success. Ivanov said that the IORT team at Celebration Health includes herself, a radiologist, a radiation oncologist and a research nurse/coordinator (Celebration Health conducts IORT as part of an ongoing study). She said that typically, the patient is referred as an IORT candidate by a primary care physician or other outside provider. The patient will then go to radiology, and proceed to the surgeon and finally the radiation oncologist to determine if they are suited for IORT. “In this day and age, unless you’re doing surgery with a team approach, you’re doing it wrong,” she said. “It’s a multidisciplinary disease, so we treat patients as a team.
“You have to look at it from a global perspective,” she concluded. “The more options you can provide a patient, the better their treatment will be.”
References
1. Schroen A, et al. “Impact of Patient Distance to Radiation Therapy on Mastectomy Use in Early-Stage Breast Cancer Patients,” Journal of Clinical Oncology, Oct. 1, 2005. Accessed Aug. 17, 2015.
2. “‘One-stop’ radiotherapy could be an alternative for early breast cancer, studies show,” The Lancet, Nov. 11, 2013. Accessed Aug. 19, 2015.


 May 07, 2026
May 07, 2026 









