Image courtesy of Varian Medical Systems.
The goal of radiation therapy is to deliver ionizing radiation to eradicate the tumor. Ionizing radiation, however, is indiscriminant and causes damage to both healthy tissues and disease volume along its path. A state-of-the-art treatment would employ intensity-modulated, irregularly shaped beams from various gantry angles to maximize the dose to the target volume and minimize the dose to the surrounding tissues. Accurate beam placement is thus critical to the success of radiation therapy.
Image-guided radiation therapy (IGRT) describes the process where imaging information acquired at the time of treatment is used to localize the target volume or its surrogates for guiding the placement of the radiation beams. Image guidance is the enabling technology that has been fundamental to the advances of modern radiation therapy. Even in its current state, IGRT continues to evolve as the community embraces more aggressive treatment and the understanding of target localization improves.
Imaging is integral to the practice of radiation therapy. The introduction of the “ready pack” films to verify beam placement in the 1970s was the precursor to image guidance. Portal images of the beam “port,” or aperture, were acquired to verify its correct placement with respect to the body anatomy. Because, by design of the conformal treatment, a portal image would contain minimal anatomic information, a double exposed image would be made to combine an open beam image of the large anatomic extent with the portal image. However, due to the relatively lengthy procedure to expose and develop the films, they were typically reviewed at the end of the treatment day and not at the time of treatment. Weekly double exposed portal imaging was the mainstay of image verification into the 1990s.
Electronic Portal Imaging Devices
The advent of electronic portal imaging devices (EPIDs) mounted onboard the linear accelerators in the 1980s helped promote the ambition of real-time IGRT. Beginning with the first-generation matrix ionization chamber system and camera-based systems, attempts were made to utilize the projection images acquired at the time of treatment to set up the patient. Contrary to the early expectation, the practice did not improve the accuracy of patient setup. It became clear that patient setup was inherently a three-dimensional (3-D) problem. Adjustment based on a single 2-D projection image would be ineffective, except for those large setup deviations where even a gross correction would be beneficial. Derivation of a more robust solution would require anatomic information from two or more distinct projections. The workflow would be changed to allow imaging and evaluation to be performed prior to treatment. These procedures deterred routine applications and were reserved for treatments that required a high degree of setup accuracy. EPIDs are nevertheless convenient replacements for port films. The digital format of the images is also highly desirable as they can be processed to improve visualization.
EPIDs made digital portal images readily available and played a vital role in shaping the practice of modern IGRT. The many studies performed on the imaging data greatly enhanced our understanding of setup uncertainties. New insight about systematic and random setup errors led to the development of various offline and online correction strategies for IGRT. In the offline approach, images from a few initial treatment sessions are analyzed to estimate the systematic error that can be corrected for subsequent treatments. Deferring image analysis away from the treatment machine offers an efficiency advantage. However, only a handful of clinics have adapted the offline approach for IGRT, as the process requires significant investment of an infrastructure for image analysis and execution of correction. In the online approach, setup errors are determined and corrected while the patient is still on the couch to be treated, accounting for both systematic and random error. Online correction using two or more pretreatment images is by far the more common practice at present. Many of the earlier impediments experienced with early EPIDs were overcome. Treatment personnel are more knowledgeable about setup variation; image quality has been improved with the modern, large area flat-panel detectors; and faster software tools for image analysis are available and are integrated with the modern digital medical accelerator to facilitate correction.
As EPIDs gained broad acceptance in the community, it became clear that the low contrast-detection capability of portal images acquired with the treatment megavoltage (MV) X-rays hampered the analysis of setup error. The significant MV dose also limited the frequency of imaging. A most significant development was the implementation in the late 1990s of a kilovoltage (kV) EPID system onboard the medical accelerator orthogonal to the treatment beam. The approach was, again, not new and was attempted in the 1950s to provide high-quality setup images on cobalt-60 treatment units. However, the more recent efforts were greatly enhanced with the availability of the flat-panel detector and modern computer technologies. Surprisingly, kV imaging was found to be no more accurate than MV imaging in determining setup error when both images contained adequate anatomic information; the reason likely due to advances in image processing. The main advantage for kV imaging laid in the reduced imaging dose that would allow more projection and more frequent imaging.
Onboard kV Imaging
The advent of onboard kV imaging coincided with the recognition that soft tissue organ displacement between treatments would impact negatively on dose escalation with the new delivery method of intensity-modulated radiation treatment (IMRT). As projection images did not contain soft tissue information, extension of onboard kV imaging for cone-beam computed tomography (CBCT) was a logical development to provide soft tissue information. Commercial kV CBCT imaging systems were launched in 2004. At present, kV CBCT is available as standard equipment on almost all medical accelerators. Onboard kV CBCT is the prevalent technology that marks the present era of IGRT.
CBCT has generated tremendous excitement by presenting the 3-D patient anatomy for treatment setup. The improved visualization has enhanced the deployment of many advanced treatment techniques such as stereotactic radiosurgery (SRS) and hypofractionated stereotactic body radiation therapy (SBRT).
There are, however, two major limitations. First, CBCT reconstructed from projection images acquired with the onboard flat-panel imager is not of diagnostic quality. The large format projection imaging has significantly more scatter contributions than conventional helical CT scanning, which degrades CBCT image quality. Similarly, the large format and slow scan speed renders the gantry-mounted CBCT system susceptible to artifacts due to patient motion. CBCT is therefore less effective in discriminating a soft tissue target in a low contrast environment. Localization of pelvic and abdominal lesions such as in the prostate, liver and pancreas is challenging with CBCT. Secondly, it is often overlooked that CBCT only provides a “snapshot” of patient information at the time of imaging, but not during the actual radiation delivery. This is a significant deficiency with lengthy SRS treatments where intra-fraction monitoring is imperative to guard against patient motion or to verify stereotactic repositioning.
Alternative Verification Methods
Alternative verification methods are used to address the limitations or complement the applications of CBCT. Detection of implanted markers on kV projection image is commonly used to localize a soft tissue target with low radiographic contrast and to monitor its motion during treatment. A radio-dense implanted marker can be localized with X-ray imaging to better than 1 mm. Ideally, three or more markers are implanted and detected by orthogonal imaging to determine the 3-D location of the centroid, or a mean position, of markers. It is generally adopted in the community for prostate localization, particularly in those clinics without in-room CT capabilities. The procedure is, however, less common for sites such as liver and pancreas where highly skilled experts are required. Even for prostate, there remains uncertainty in equating the mean position of the implanted markers to that of the target. Agreement between corrections based on the prostate volume in CBCT with those based on implanted markers is modest. A study at the Mayo Clinic shows that about 25 percent of the corrections based on CBCT differ from those based on markers by more than 5 mm. Longitudinal MRI studies indicate that the positional correlation between markers and the prostate is a function of implanted marker positions. It follows that the optimal positions for implanting markers and the corresponding setup margins for different disease sites require more rigorous validation for motion management and IGRT.
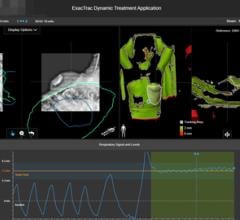
Issues of implant positions notwithstanding, implanted markers do offer the significant advantage of monitoring intra-fraction motion during radiation delivery. Fluoroscopic imaging can be used, albeit with careful consideration of imaging dose. Of significant impact to marker-based guidance is the revolutionary non-ionizing electromagnetic (EM) technology that aptly addresses the challenge of intra-fraction monitoring. Varian’s Calypso system provides continuous real-time detection of the 3-D location of implanted EM transponders, or beacons, during treatment. Its localization performance is impressive and rivals that of the kV X-ray imaging technique. To date, its clinical application has been limited to the prostate. A major impediment to the EM approach is the use of a relatively large needle to implant each 1.8 mm wide and 8.5 mm long beacon. The incompatibility of the beacons with MRI also significantly hinders the use of MRI for treatment planning or assessment.
Current Challenges
At present, the challenge of soft tissue target localization with CBCT continues to spur the development of other innovative anatomic image guidance approaches for IGRT. There is increasing use of room-mounted optical systems to guide treatment setup and monitoring where the patient surface images are used as surrogates for internal anatomies. Another technology that is undergoing a revival is the use of ultrasound (US) imaging for soft tissue localization in the abdomen. The introduction of intra-modality verification where the US images acquired at time of CT simulation is used to register with and localize the US target at time of treatment has lessened the concern for subjective image interpretation. Most notable are the ongoing development of medical accelerators with onboard MRI. The combination is very powerful, although it is also expected to be more costly and requires significant adjustment of clinical infrastructure and workflow. All these technologies are highly attractive as they are nonionizing and noninvasive to facilitate continuous monitoring of the patient while the beam is on. The investigations into intra-fraction monitoring of soft tissue perhaps mark a new era in IGRT. Care must be taken to validate the imaging information for optimal clinical application. As demonstrated so many times in the past, imaging is fundamental to the advances in radiation therapy. itn
John W. Wong, Ph.D., is professor and director of the division of medical physics in the department of radiation oncology and molecular radiation sciences at Johns Hopkins University School of Medicine.


 February 23, 2026
February 23, 2026