May 20, 2015 — Adding the HeartFlow FFRCT Analysis to a standard coronary computed tomography angiogram (cCTA) may change the course of treatment in more than one-third of patients with coronary artery disease. This conclusion was discussed in a study presented at the EuroPCR 2015 conference.
Results of the FFRCT RIPCORD study, presented by Prof. Nick Curzen of the University Hospital Southampton, United Kingdom, further reinforce the ability of the HeartFlow Analysis to aid in patient management.
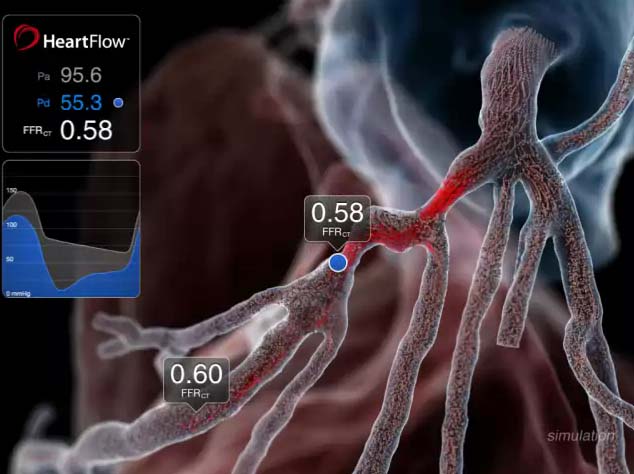
The HeartFlow Analysis is a non-invasive imaging technology for coronary artery disease offering physicians insight on both the extent of any arterial blockage as well as whether it is impacting blood flow — two vital pieces of information physicians need to develop a treatment plan that is right for a patient. Using standard cCTA images, HeartFlow’s technology solves millions of complex equations simulating blood flow in the coronary arteries to provide mathematically computed fractional flow reserve (FFRCT) values.
In the study, three experienced cardiologists reviewed cCTAs of 200 consecutive cases of patients with stable chest pain. The physicians agreed upon a plan to manage each patient: optimal medical therapy, a percutaneous coronary intervention (PCI) such as a stent or coronary bypass surgery. In some cases, the physicians agreed that they were unable to make a decision and needed more information, such as an invasive diagnostic angiogram with associated invasive FFR.
Next, the physicians were shown the results of the HeartFlow FFRCT Analysis for each case and made a second management decision incorporating these data. Adding the HeartFlow Analysis changed the decision to medical therapy in 26 of the 87 cases (30 percent) that were originally recommended for a PCI. Of cases originally recommended for medical therapy, 7 of 67 (9 percent) were changed to PCI and one was changed to bypass surgery. Without the HeartFlow Analysis, the physicians needed more information in 38 cases (19 percent), but with the HeartFlow Analysis, there were no cases in which the physicians needed additional information.
Overall, the management plan was changed in 72 of the 200 patients (36 percent). In 16 of the 87 cases considered candidates for PCI based on the angiogram (18 percent), one or more of the target lesions was changed using the FFRCT Analysis.
“It is well established that invasive FFR can refine management decisions based solely upon the angiogram for patients with chest pain,” said Curzen. “This study demonstrates that the non-invasive HeartFlow FFRCT Analysis has the potential to become the default method for the initial assessment of many patients with cardiac-sounding chest pain by assessing both the coronary anatomy and physiology simultaneously. This could have important implications for our clinical practice and would challenge conventional care pathways.”
Invasive FFR is seen as the gold standard in diagnosing functionally significant coronary artery disease. In patients with multi-vessel disease, an FFR-directed PCI strategy is associated with better clinical outcomes compared to angiography-directed PCI (FAME; FAME 2). The FFRCT RIPCORD study was modeled after the original RIPCORD study, in which invasive FFR altered the course of patient management in 26 percent of cases when compared to angiographic assessment alone.
For more information: www.heartflow.com


 May 27, 2026
May 27, 2026 









