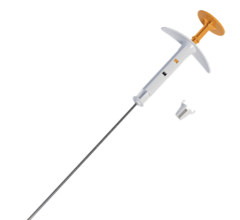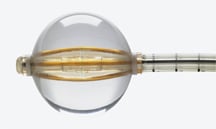
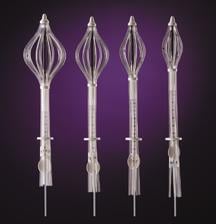
SenoRxs Contura is a multilumen radiation balloon applicator for accelerated partial breast irradiation.
In the war against breast cancer, brachytherapy is a powerful technique for eradicating the spread of the disease. However, certain limitations in the method of delivering radiation have excluded a large patient population that would benefit from the treatment.
To overcome this barrier, researchers are developing customized brachytherapy techniques that afford the flexibility for physicians to deliver personalized care with hopes of saving more lives.
A Simple, Single Solution
Until recently, the availability of breast brachytherapy has been limited to only a small number of women due to the way it uniformly distributes radiation. The uniform dispersement of radiation does not allow the physician to customize the radiation delivery, and as a result exposes healthy tissue over a longer period of time. This technicality makes it difficult for women with smaller breasts or those with minimal space between the balloon and their skin to receive partial breast radiation treatment.
However, new breast brachytherapy tools that allow physicians to customize the radiation dose to different areas of the lumpectomy cavity are expanding the pool of women who are now able to receive such treatment, thereby decreasing side effects and expediting treatment. One such tool, the SAVI device developed by Cianna Medical Inc., is a single-entry, interstitial-type brachytherapy applicator implemented as part of breast conservation therapy.
According to a recent study led by Dan Scanderbeg, Ph.D., which was published in Radiotherapy and Oncology, the SAVI applicator enables more women to receive breast brachytherapy. The study involved women who had a distance of 7 mm or less between the device and the skin, which consisted of approximately 40 percent of the women in the study. This skin separation distance prevented them from receiving balloon brachytherapy. However, the study found that SAVI had the capability of directing the radiation dosage away from the skin, which enabled this group of women to receive this type of breast brachytherapy.
“The single-entry, multicatheter SAVI applicator is both flexible and easy to use,” said Catheryn Yashar, M.D., assistant professor and chief of breast and gynecological services, University of California-San Diego and co-author of this study. “It is the only single-entry device that allows physicians to customize radiation based on the size and shape of the cavity. SAVI spares healthy tissue while directing radiation to where it is needed most. That can make brachytherapy available to women who would otherwise be ineligible for such treatment, when the required treatment involves radiation areas that lie in close proximity to the sides of the breast,” Dr. Yashar indicated. “There is no contest [with other accelerated partial breast irradiation devices] in tight spaces.”
Patient convenience and comfort are other advantages of the SAVI applicator. According to Dr. Yashar, pain has not been a complaint from any of her patients. The catheter is inserted using local anesthetic and has been reportedly painless to remove. Additionally, with SAVI radiation treatment can be completed in just five days as compared to six weeks for whole breast radiation.
Dr. Yashar’s experience with SAVI has also included using the device on more challenging patient situations, such as those with irregularly shaped lumpectomy cavities. Because most cavities are not perfectly spherical, many women are not candidates for traditional balloon brachytherapy. However, this tool is said to be effective even with irregularly shaped cavities and can radiate areas as small as 5 to 10 cubic cm.
The manufacturer also recently introduced the 6-1 Mini, which is the smallest SAVI device and is designed to treat the smallest cavities, including ones in the upper inner quadrant of the breast.
Multipronged Approach
Another similar device, Contura, developed by SenoRx Inc., has also eased the barrier to entry to include more women who may receive partial breast radiation. Contura is a multilumen radiation balloon applicator, which uses a vacuum to remove excess fluid and to adhere closely to irregularly shaped cavities.
According to Kenneth Tokita, M.D., radiation oncologist at the Cancer Center of Irvine, CA, there are two main advantages of the Contura that set it apart from other breast brachytherapy instruments. These include the construction of the device consisting of five catheters within a single balloon, which enables him to better shape the radiation field, and the ability of the device to suck out air and blood from the cavity before radiation. He explained that the ability to contour the field of radiation is much improved; with previous tools, only about one-third of his patients were not candidates for such treatment due to the radiation field being too close to the lungs or ribs.
In February of this year, SenoRx expanded its labeling for its larger-size Contura which indicates its suitability for lumpectomy cavities ranging from 4.5 to 6 cm in comparison to its previous labeling of 5 to 6 cm. Although the device is geared for this range, Dr. Tokita has used Contura to radiate cavities ranging from 2 to 2.5 cm to about 4.5 cm.
“So, you can do quite a range. Contura will cover about 95 percent of all lumpectomies for localized breast cancer,” said Dr. Tokita. According to him, the 5 percent that are not candidates for Contura are patients with very large lumpectomy cavities, larger than 4.5 to 5 cm. “It’s pretty rare to see one smaller than 2 cm by the time the surgeons take the tumor out and get a margin. I don’t know that I’ve ever seen one smaller than 2 cm.”
When asked how he thinks Contura stacks up against other similar devices, such as SAVI, Dr. Tokita explained, “[With SAVI] the radioactive material is right on the surface of the inner cavity. So when you twist the head, it expands out to fill the cavity. In that sense, you can control the contour of the radiation better than you can with Contura, but the radiation is very hot, so you get some real hot spots along the inner aspect of the cavity. I could not in my own mind make a real clean radiation without extreme hot spots along the cavity edge, which would really cut down the healing.”
Since long-term infection is a major concern for partial breast radiation, one benefit of Contura is its ability to remove blood and air from the cavity, which diminishes the risk of infection and other complications. Out of the 65 cases conducted so far, only two patients have developed infections, each of which was easily resolved.
According to Dr. Tokita, other side effects include pain one or two days after the device is implanted into the breast and discomfort in some patients upon removal.
Overall, Dr. Tokita said, “To be honest with you, right now I don’t know how they could improve it. It’s really a pretty impressive piece of equipment.”
Brachytherapy Put to the Test
One of the leading brachytherapy balloons is MammoSite by Hologic Inc., a leading women’s healthcare device manufacturer. The MammoSite balloon is placed inside the lumpectomy cavity through a small incision in the breast through a catheter. During therapy, a portion of the catheter that remains outside the breast is connected to a computer-controlled High Dose Rate system that inserts a radiation seed. The treatment lasts five days.
One of the significant advantages of the device is its testing track record. In a five-year study, MammoSite proved comparable to long-term interstitial brachytherapy and whole breast radiation therapy for both recurrence rates, and cosmetic results were above 80 percent.
In another study, presented by Peter Beitsch, M.D., of Medical City Dallas Hospital at ASTRO 2008, researchers conducted a four-year analysis on patients enrolled in the American Society of Breast Surgeons MammoSite Breast Brachytherapy Registry Trial.
Their analysis of the overall registry population with a median follow-up of 36 months showed a three-year actuarial rate of ipsilateral breast tumor recurrence (IBTR) of 2.15 percent. Other follow-up results were also positive — 0.26 percent three-year actuarial rate for axillary recurrences and 0.74 percent three-year actuarial rate for new primary cancers.
In a subset of patients (the first 400 treated), 44-month follow-up data was captured and a 93.9 percent survival rate was reported (88.9 percent disease-free and 100 percent cancer-specific). The researchers concluded that treatment with the MammoSite system resulted in low IBTR, low axillary recurrence and acceptable cancer specific survival.
There is no doubt that there are several approaches and instruments to effectively deliver breast brachytherapy, and having those options allows doctors to offer each patient more personalized treatment.
Each one of these instruments, Mammosite, SAVI and Contura, provide those options and have opened the doors for many more women to receive accelerated partial breast radiation treatment.


 October 01, 2023
October 01, 2023