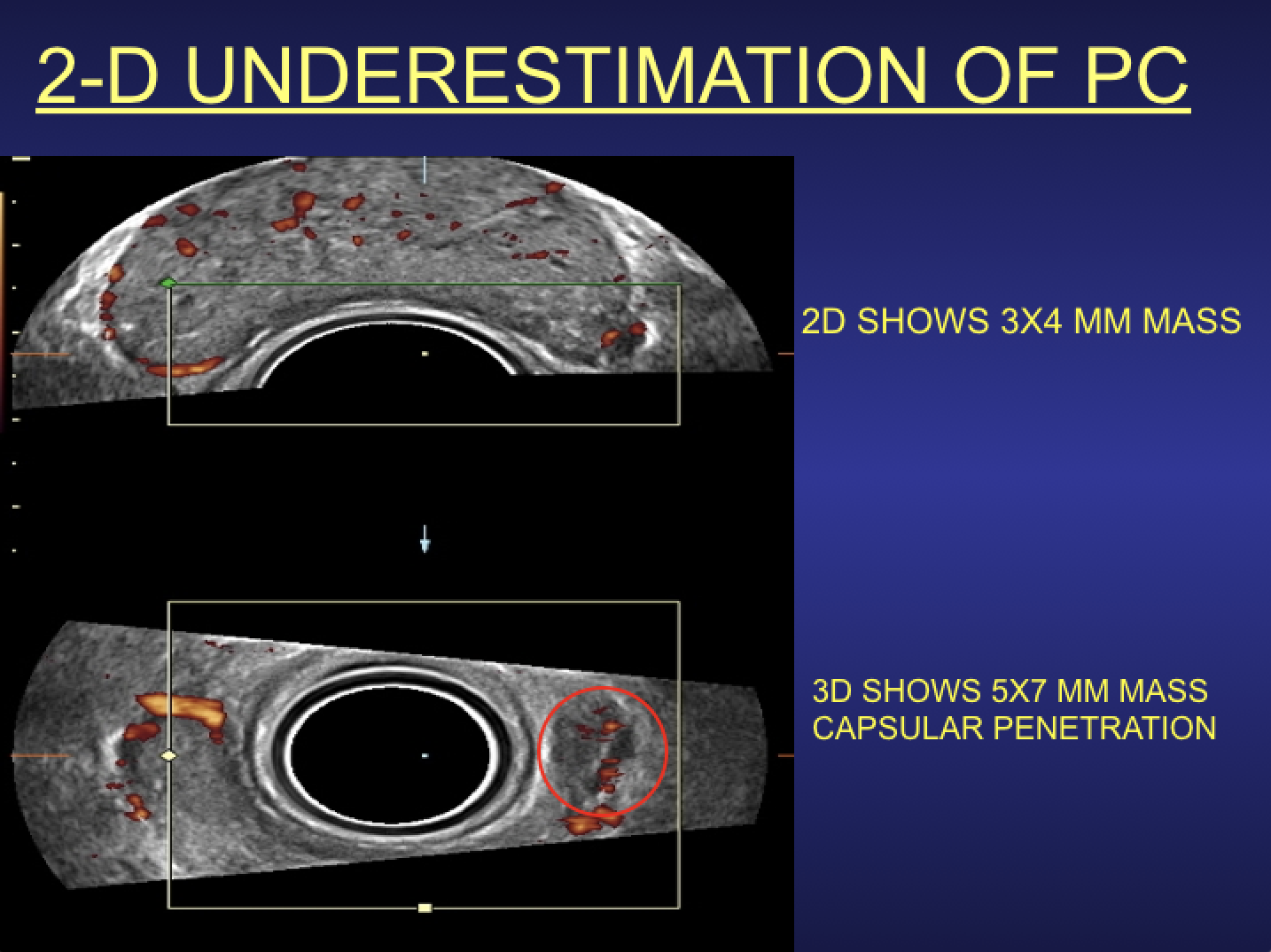
Figure 1. 2-D (top) vs 3-D (bottom). The irregularly marginated left tumor mass that bulges the lateral capsule appears three times the volume on the multiplanar 3-D/4-D image reconstruction and shows complete extraprostatic extension. Note: This anterolateral lesion is not clinically palpable.
Extracapsular extension (ECE) is a major clinical indicator for brachytherapy or external beam treatment of prostate cancer. The prostatic capsule is a thin layer of compressed fibrous tissue and ultrasound imaging depicts the pericapsular fat as a strongly echogenic structure while magnetic resonance imaging (MRI) demonstrates a dark envelope. The capsule tends to be incomplete around the apex accounting for greater extraprostatic spread from apical tumors. Applying standard diagnostic criteria, capsular erosion must appear in two orthogonal views or in multiplanar 3-D volumetric sonography 2/3 planes. Unlike standard ultrasound where the operator moves the probe, volumetric 3-D transducers are stationary as 200-300 images are captured in 10 seconds, removing the problem of operator dependency and allowing rapid review and accurate comparison of pathology in subsequent studies. (See Figure 1.)
Capsular invasion occurs with infiltration of the cancer into the fibrous capsule. Capsular penetration is tumor invasion extending beyond the capsule that produces bulging, irregularity of the capsule and gross tumor noted invading the fascial planes, rectal serosa or levator sling muscles. Perineural invasion, a microscopic diagnosis, cannot be inferred from diagnostic imaging at this time. However, the nerve supply of the gland tends to follow the major arterial network and is imaged with color Doppler sonography (CDS) or power Doppler sono-angiography (PDS). Capsular lateral wall erosion is often best imaged in the coronal view on 3-D PDS, while posterior erosion usually is best depicted on the transverse 3-D PDS. MRI, with its inferior capsular resolution (including endorectal coil scans) may not always confirm extracapsular disease clearly exhibited on high-resolution sonography.1-7 During examination of a suspicious capsule finding, the real time 4-D mode might be used to target the anomaly and better image the full anatomic extent of penetration with curvilinear probes.
Quantifiable Digital Scanning Versus Biopsy
The new optical dermatologic modalities of reflectance confocal microscopy (RCM) and optical coherence tomography (OCT) are highly accurate in detecting malignant capsular penetration and are used in other solid organ tumors such as breast, thyroid and prostate tumors with great accuracy, but limited depth penetration. Highly suspect areas are then checked for spread to the seminal vesicles, and a search is performed for lymphadenopathy to verify the disease is locoregionally confined and surgical intervention is unlikely at this time. Patients are reassured since they simultaneously see the 3-D picture as the real time exam proceeds in thoroughly logical, systematic staging. Four-dimensional sequencing (real time 3-D) permits image-guided biopsy of the most virulent area of the tumor and allows the pathologist to focus on strongly suspicious regions of the lymph node mass excised during prostatectomy. Similarly, during examination the patient may personally visualize extraprostatic disease or seminal vesical invasion so plans for radiation or other therapies may be initiated in a timely manner.
Digital Imaging Reduces Complications
Fear of complications deters patients from seeking medical intervention so many opt for noninvasive diagnostic options. Cancer treatment effectiveness depends on the precise tumor volume and depth penetration measurements. However, many cancers provoke a benign local immune response or there may be co-existant inflammatory reaction that simulates a larger area of malignancy, such as cicatrix formation accompanying chronic prostatitis, that is indistinguishable on MRI from a stone or malignancy. Imaging highlights the true border of the tumor, optimizing targeting for healthy tissue sparing and resulting in a narrower treatment zone and lesser side effects.
Doppler Applications
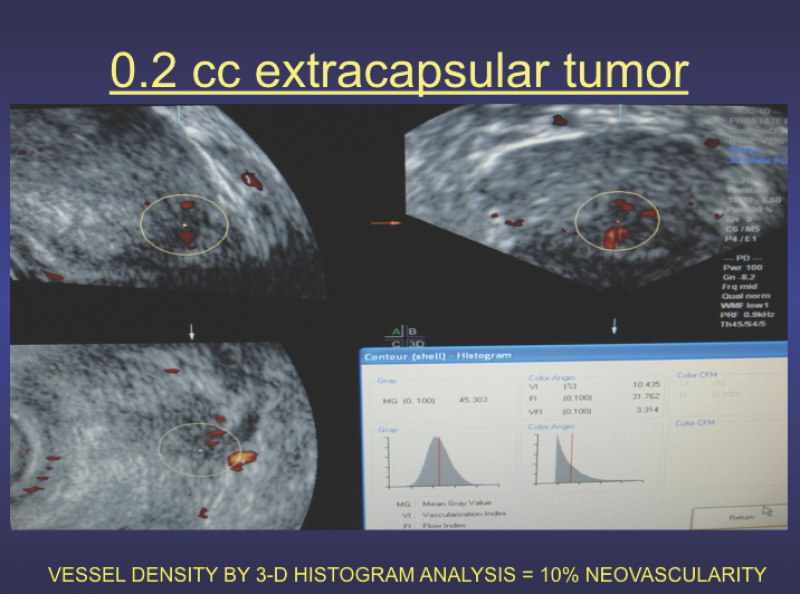
Blood vessel mapping using the various Doppler modalities is routinely used in both cancer treatment and reconstructive preoperative planning. In cancer radiation, the presence of aberrant large veins or significant arteries in the treatment field may allow post-treatment vasculitis to be minimized. Advanced 3-D Doppler systems allow for histogram vessel density measurement of neoplastic angiogenesis. (See Figure 2.) This baseline of neovascularity is used worldwide as a reliable treatment parameter and serves as an accurate surrogate endpoint.8-22
Low grade prostate cancer identified by 4-D volumetrics clearly delimited by the capsule and of low tumor vessel density is followed serially in 6-month intervals. Subsequent capsular invasion or increase in vessel density by histogram analysis requires urologic or oncologic measures. Primary bladder cancer or extracapsular extension into the bladder is evaluated concomitantly with the prostate and neovascularity and wall invasion noted before surgical referral. Rectal and bladder post-radiation effects may be moderated by pretreatment placement of tissue buffering devices under ultrasound image guidance.
Contrast Enhanced Ultrasound (CEUS)
In 1990 Rodolfo Campani, M.D., director of the University of Pavia Radiology Department, developed ultrasound contrast-enhanced cancer imaging for liver tumors followed by David Cosgrove, M.D. (London), Nathalie Lassau, M.D. (Paris) and Carlo Catalano, M.D. (Naples), for breast, prostate and melanoma respectively. CEUS is currently used worldwide, but not fully FDA-approved in the United States. Microbubble media show tumor neovascularity with exquisite detail, evaluating therapeutic response in solid organs. This distinction is important since the international response assessment in solid tumors (RECIST) studies demonstrated tumor enlargement during treatment may be related to apoptotic cell death with cystic degeneration or immune cell infiltration destroying malignant tissue while increasing the visualized tumor volume. Doppler ultrasound or CEUS reliably verifies decreased malignant angiogenesis in these cases instead of using biopsy, contrast computed tomography (CT) or DCE-MRI for confirmation. Thermal treatments such as cryotherapy, high-intensity focused ultrasound (HIFU) or laser ablation are designated complete when penetrating cancer arteries are no longer detectable. It must be noted that prostatitis and post-treatment changes by radiation or other therapies distorts the expected imaging anatomy by most modalities, and the true evaluation of recurrence is best determined by functional vascular perfusion studies that are superior surrogate tumor markers to other clinical indicators. A recently recognized immuno-oncologic response limiting positron emission tomography (PET)/CT accuracy is termed “pseudo-progression” demonstrating overall tumor burden increases during successful therapy. Shrinking tumors may likewise be biologically viable as is occasionally seen from the aggressive desmoplastic reaction to certain high-grade breast cancers. Post radiotherapy imaging in successful cases may show decrease in tumor neovascularity in as short a time period as 1-2 weeks, suggesting short interval neovascular evaluation as a better indicator of effective response than standard protocols. In the future, ionizing treatments may be considered effective when tumor vessel perfusion has completely ceased at a 3-month scan interval and further biopsies possibly obviated. Post-radiation prostate-specific antigen (PSA) spikes are quickly shown to be false positives due to prostatitis or bone metastases rather than actual recurrence in the treatment glandular area.
Margin Delineation
Advances in laser/optical devices allow near microscopic tissue analysis of the cells by noninvasive testing. Real time microscopy during surgery verifies the tumor border is clear in cases of skin cancer, and future use in breast and prostate cancer treatment is under clinical study. In true capsular penetration, radiation treatment becomes a prime clinical option. Since the 18 MHz probe is directly in contact with the capsule, the resolution is five times greater than MRI capability. DCE-MRI is routinely used to verify malignant lymphadenopathy or bone metastases, and has been highly successful after 3-D/4-D imaging has ascertained and localized the extraprostatic disease.23 Another potential use of margin neovascularity is the problem with anatomic pathologists seeing biopsy specimens that appear malignant but not being able to assess if the microscopically abnormal cells are biologically active. Some of the newer optical applications image capillary blood flows, meaning the total absence of microcirculation may signify a treated tumor under the microscope may look active, but be functionally inactive at this time.
Autoimmune Disease, Toxic Exposure and Cancer
Abnormal immune responses that initially appear in the skin are associated with increased multiorgan cancer incidence including breast and prostate disorders. Inflammatory vessels in psoriasis and infection are visibly observed and quantifiably cataloged since successful treatment is calibrated by measured decrease in the number and type of abnormal vessels. High vascular immune vessel density is proportional to increased risk of future neoplastic tissue proliferation. Many arthritic conditions have coexistent dermal manifestations alerting us to the probability of more extensive subclinical joint involvement. Thus patients with proven collagen disorders, contact dermatitis of unknown etiology or overexposure to toxins as we see in the 9/11 first responders are candidates for cancer screening at 6 month intervals. The statistics from the cancer organizations monitoring the World Trade Center disaster have found the incidence of skin cancers to be highest followed by bladder and prostate cancer. The general public is sensitized to the fact that our contaminated food supply, medicines and overall environment has increased our risk and are demanding more accurate cancer detection options.24 A study by William Oh, M.D., reported in Business Insider, June 2019, done at Mt Sinai on the firefighters showed a surprising result that toxic dust inhalation produced more prostate cancers than lung cancers in the Ground Zero responders.25 Clearly more attention needs to be paid to environmental carcinogens and increased cancer screening will discover more disease at a lower stage, improving overall survival.
Since 1974, Robert L. Bard, M.D., PC, DABR, FASLMS, has pioneered noninvasive digital imaging technologies as alternatives to radiation-producing diagnostic systems for evaluating solid organ neoplastic disease. He is internationally recognized as a leader in the field of 21st century 3-D ultrasonographic volumetric Doppler imaging.
He specializes in advanced 3-D sonography to detect cancers in numerous organs including the breast, prostate, skin, thyroid, melanoma and other areas. His images are used to accurately guide biopsies, target therapy and provide focused follow-up after treatment.
References
1. Neal DE, Donovan JL. Prostate cancer: to screen or not to screen? Lancet Oncol 2000;1(1):17-24.
2. Resnick NI, Willard JW, Boyce WH. Ultrasonic evaluation of the prostatic nodule. J Urol 1981;120:86-9.
3. Rifkin M, Sudakoff G, Alexander A. Prostate: techniques, results and potential applications of color Doppler US scanning. Radiology 1993;186:509-13.
4. Aarninck RG, Beerlage HP, De La Rosette JJ., Debruyne F.M, Wijkstra A. Transrectal ultrasound of the prostate : innovations and future applications. J Urol 1998;159:1568-1579.
5. Kelly I, Lees W, Rickards D. Prostate cancer and the role of color Doppler US. Radiology 1993;189:153-6.
6. Newmann JS, Bree RL, Rubin JM. Prostate cancer: diagnosis with color Doppler sonography with histological correlation of each biopsy site. Radiology 1995; 195(1):86-90.
7. Bree RL. The role of color Doppler and staging biopsies in prostate cancer detection. Urology 1997;49:31-4.
8. Lavoipierre AM, Snow RM, Frydenberg M, Gunter D, Reisner G, Royce PL, Lavoipierre GL. Prostatic cancer: role of color Doppler imaging in transrectal sonography. AJR 1998;171(1):205-10.
9. Cornud F, Belin X, Piron D, Chrétien Y, Flam T, Casanova JM, et al. Color Doppler-guided prostate biopsies in 591 patients with an elevated serum PSA level: impact on Gleason score for nonpalpable lesions. Urology 1997;49(5):709-15.
10. Cornud F, Hamida K, Flam T, Hélénon O, Chrétien Y, Thiounn N, et al. Endorectal color doppler sonography and endorectal MR imaging features of nonpalpable prostate cancer: correlation with radical prostatectomy findings. AJR Am J Roentgenol 2000;175(4):1161-8.
11. Halpern EJ, Strup SE. Using gray-scale and color and power Doppler sonography to detect prostatic cancer. AJR 2000; 174:623-627.
12. Sauvain JL, Palascak P, Bremon JM. Power Doppler ultrasonography and hypoechoic nodules of peripheral prostate: prospective and limitations. J Radiol 1997;78:491-497.
13. Kuligowska E, Barish MA, Fenlon HM, Blake MA. Predictors of prostate carcinoma: accuracy of gray-scale and color Doppler US and serum markers. Radiology 2001;220:757-64.
14. Sauvain JL, Palascak P, Nader N, Gomez W, Bloqueau P, Bremon JM, et al. Echo-Doppler et cancer de la prostate: option ou nécessité? J Radiol 2006;87(9):1063-72.
15. Sauvain JL, Palascak P, Bourscheid D, Chabi C, Atassi A, Bremon JM, et al. Value of power doppler and 3D vascular sonography as a method for diagnosis and staging of prostate cancer. Eur Urol 2003;44(1):21-30.
16. Sauvain JL, Sauvain E, Rohmer P, Louis D, Nader N, Papavero R, Bremon JM, Jehl J. Value of transrectal power Doppler sonography in the detection of low-risk prostate cancers. Diagnostic and Interventional Imaging (2013)94:60-67.
17. D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998;280:969-74.
18. D’Amico AV, Chen MH, Roehl KA, Catalona WJ. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N Engl J Med 2004;351:125-35.
19. Dall’Era MA, Konety BR, Cowan JE, Shinohara K, Stauf F, Cooperberg MR, et al. Active surveillance for the manage- ment of prostate cancer in a contemporary cohort. Cancer 2008;112(12):2664-70.
20. Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA 2005;293:2095-101.
21. Stoica G, Cariou G, Colau A, Cortesse A, Hoffmann P, Schaetz A, Sellam R. Epidemiology and treatment of acute prostatitis after prostatic biopsy. Prog Urol 2007 Sep;17(5):960-3.
22. Bard R. 2009 Dynamic Contrast Enhanced MRI of Prostate Cancer. Springer Heidelberg
23. Bard R Ed 2014 Image Guided Prostate Cancer Treatments Springer New York
24. Bard R Ed 2019 Image Guided Dermatologic Treatments Springer Berlin
25. https://amp.businessinsider.com/what-causes-prostate-cancer-911-responders-new-clues-2019-6



 April 30, 2026
April 30, 2026