ProHance is a macrocyclic, nonionic, gadolinium-based magnetic resonance imaging (MRI) contrast agent designed for use in MRI in adults and children over two years of age to visualize lesions with abnormal vascularity in the brain (intracranial lesions), spine and associated tissues. ProHance is also indicated for use in MRI in adults to visualize lesions in the head and neck.
June 9, 2008 - Siemens unveiled the ACUSON SC2000 volume imaging ultrasound system, which acquires nonstitched, real ...
The FDA granted tentative approval for Covidien’s Abbreviated New Drug Application (ANDA) for its Kit for the Preparation of Technetium Tc 99m Sestamibi Injection. Covidien’s tentatively approved product is a generic of Cardiolite, which is a myocardial perfusion imaging agent used for detecting coronary artery disease.
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
June 9, 2008 – American TeleCare Inc. (ATI) released Quick Notes as a new feature of its inLife/LifeView Telehealth ...
The FDA recently approved Covidien’s contrast delivery system with radiofrequency identification (RFID) technology ...
The EmpowerMR injector system has multiple features created to cope with the problem of electrical interference in the ...
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
The Deficit Reduction Act of 2005 (DRA), which went into effect on January 1, 2007, has left its mark on hospitals and ...
June 9, 2008 - Cardinal Health today launched a new system that leverages the company’s dispensing technologies to ...
GE received FDA clearance for its new Signa MR750 3.0T scanner that delivers up to 60 percent additional anatomical ...
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
The ACIST CVi Contrast Delivery System provides one injection system for all cardiac and vascular angiography procedures. The variable-rate ACIST CVi system aims to provide ease of operation for clinicians, while obtaining quality images with less contrast used and delivered to the patient.
June 9, 2008 - Siemens Medical Solutions gained FDA 510(k) market clearance for the SOMATOM Definition AS, what is said ...
The FDA cleared GE Healthcare’s new LightSpeed CT750 HD, a high-definition CT scanner that is said to produce images 100 ...
eHealth Saskatchewan plays a vital role in providing IT services to patients, health care providers, and partners such ...
The Codonics Virtua Medical Disc Publisher is designed to set a new standard for speed, efficiency and ease of use in ...
The future is looking bright for virtual colonoscopy (CTC) after encouraging news came out that the Centers for Medicare ...
June 9, 2008 - Curlin Medical was recently awarded a three-year contract to provide its advanced pain management-local ...
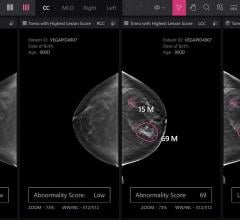
iCAD’s SecondLook Digital Computer-Aided Detection system for mammography received FDA clearance for sale with FUJIFILM ...
Bayer HealthCare Pharmaceuticals collaborated with Mobile Aspects to create VistaTrak, a touchscreen contrast media ...
June 9, 2008 - The FDA cleared Mindray’s DC-3 color ultrasound imaging system, equipped with extensive applications in ...
June 9, 2008 – Electronic health records maker MediNotes Corp. said it added 199 new software agreements with new and ...
Viztek introduced fully equipped mobile imaging vehicles, which includes a Honda Element equipped with Viztek Opal-RAD PACS and Kodak Point-of-Care CR, providing radiology groups with a turnkey solution to broaden their patient base, also making diagnostic imaging accessible in impoverished areas.

 June 08, 2008
June 08, 2008