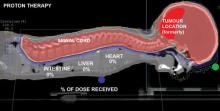
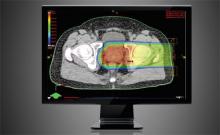
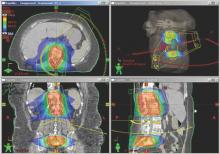
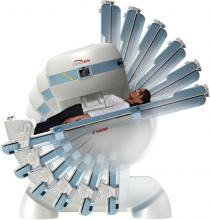
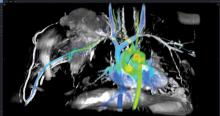
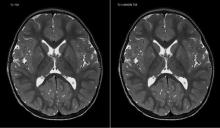
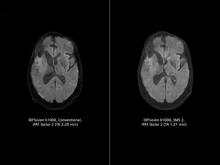
As proton therapy becomes more widely available, imaging for the development of proton treatment plans will become more commonplace. However, there are major differences in how treatment is planned for proton therapy compared to traditional radiation therapy. While anatomical imaging is the main focus in radiation therapy, proton therapy requires highly calibrated computed tomography (CT) datasets that are used to directly compute a highly technical treatment plan.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now