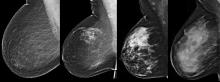
Breast density reporting is here to stay. Nineteen states have already enacted mandatory density inform laws, and additional states will continue to draft and pass bills. At the federal level, a national standard for breast density notification in the form of a Mammography Quality Standards Act reporting amendment is likely either through legislation or regulation. For clinicians practicing in states with breast density inform laws in effect, or imminent, the next practical concern is clinical staff preparedness to address patient questions.
If you enjoy this content, please share it with a colleague
- Read more about Breast Density: Are You Informed?
- Log in or register to post comments