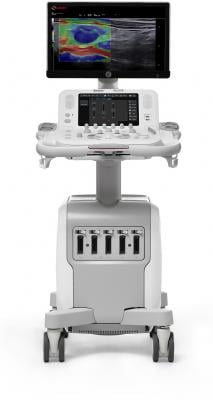
January 20, 2021 — Esaote North America announces that the MyLab X8 Ultrasound System has now been approved in Canada.
Previously approved by FDA in the USA, MyLab X8 expands the reach of the MyLab Ultrasound Product Line with a fully featured premium imaging solution, integrating the latest technologies and delivering superior image quality without compromising workflow or efficiency.
“The MyLab X8 Ultrasound System is uniquely suited for the demands of a myriad of applications, whether in the hospital or in the office,” said Robert Lewis, President and General Manager at Esaote North America. “Esaote has a legacy of producing high quality imaging solutions that are accessible and incorporate advanced technologies. We are committed to developing fully featured and versatile imaging systems with unmatched efficiency and scalability.”
Esaote has always invested in workflow-oriented design and collected several awards about innovative solutions, setting itself as one of the world’s leading providers of diagnostic imaging solutions. MyLab X8 Ultrasound System includes two exclusive solutions, easyMode and easyColor, which simplify the number of parameters to be adjusted by the users and drive ultrasound image optimization in just 3 swipes, with improved efficiency and patient throughput as a result.
A wide range of cutting-edge technologies is also available as options on MyLab X8: Virtual Navigator for fusion imaging, QElaXto shearwave elastography, very High-frequency probes as well as several automation tools like eScan, eDoppler and zero-click advanced functions.
For more information: www.esaote.com


 May 07, 2026
May 07, 2026 









