November 30, 2019 — To help empower clinicians and patients in their fight against breast cancer, GE Healthcare introduced Serena Bright, the healthcare industry’s first contrast-enhanced mammography solution for biopsy, at RSNA19.
Interventional and biopsy procedures are a necessary step in most breast cancer diagnoses. Previously, for lesions not seen on mammography or ultrasound, contrast-enhanced biopsy was typically performed with breast MR, which can be more time-consuming, uncomfortable and costly for patients. With Serena Bright, breast biopsy exams can now be done with the same mammography equipment and in the same room as the screening or diagnostic mammogram. This can help save time, lessen costs and ultimately help improve the patient experience.
“This new mammography-guided biopsy technique offers an alternative to MR-guided breast biopsy, which can improve the patient experience and allow us to get answers to patients faster,” said Corinne Balleyguier, M.D., head of medical imaging at Gustave Roussy in France. “We are proud to be the first in the world to be able to offer this technology to our patients and believe this will have a significant impact on breast imaging capabilities.”
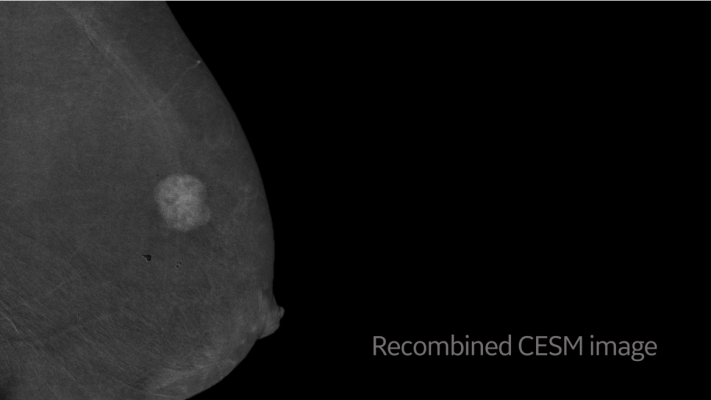
Key to this revolutionary change in breast care is GE Healthcare’s SenoBright HD Contrast Enhanced Spectral Mammography (CESM), a diagnostic imaging tool that can detect malignant lesions not seen on mammography or ultrasound in certain cases. By highlighting areas of unusual blood flow to help localize lesions that need to be biopsied, CESM biopsy — Serena Bright — helps improve radiologists’ diagnostic confidence with the ultimate goal of getting answers to patients as quickly as possible.
“This novel approach has the potential to be a more affordable and faster alternative to MR-guided biopsy for suspect findings seen in CESM,” said Rodrigo Alcantara, M.D., head of breast imaging section at Hospital del Mar in Spain. “Our initial findings have been incredibly successful in reliably targeting and enhancing lesions to ultimately find cancer earlier.”
“We are thrilled to introduce this industry-first solution to equip clinicians with another key tool in the fight against breast cancer,” added Agnes Berzsenyi, president and CEO of Women’s Health at GE Healthcare. “Through a multi-modality and personalized approach, our hope is that we can find cancers earlier to ultimately improve outcomes and save lives.”
* 510k pending at FDA. Not available for sale in the United States.
For more information: www.gehealthcare.com


 May 22, 2026
May 22, 2026 









