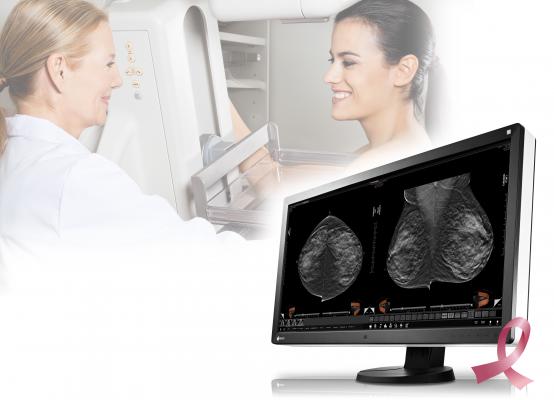
May 4, 2016 — Eizo Inc. recently announced that it received U.S. Food and Drug Administration (FDA) 510(k) clearance for breast tomosynthesis for its 8 megapixel multimodality monitor, the RadiForce RX850.
Digital breast tomosynthesis is a method of breast screening that allows the radiologist to view detailed three-dimensional images of the breast assembled from multiple X-ray pictures. When used in combination with digital mammography, tomosynthesis provides radiologists with multiple screening techniques that can be utilized together for increased diagnostic precision in breast cancer detection. The RadiForce RX850 now has FDA 510(k) approval in tomosynthesis, mammography and general radiography.
The RadiForce RX850’s super high-resolution screen (4096 x 2160) displays 8 megapixels of information with a pixel pitch of 0.1704 mm for viewing medical breast images in exceptional detail. To detect the smallest structures, the monitor offers a high contrast ratio of 1450:1. The deeper black levels distinguish similar shades of gray for sharper monochrome image reproduction.
For more information: www.eizo.com


 May 07, 2026
May 07, 2026 









