Images courtesy of Philips
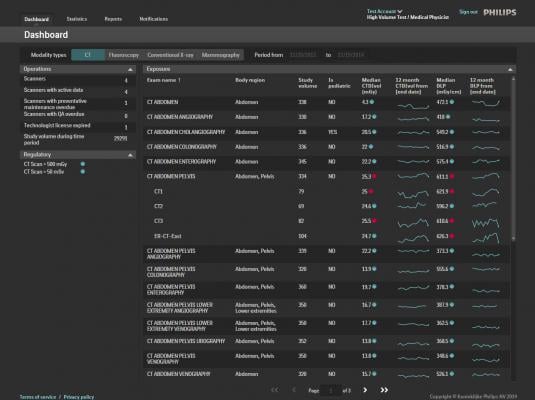

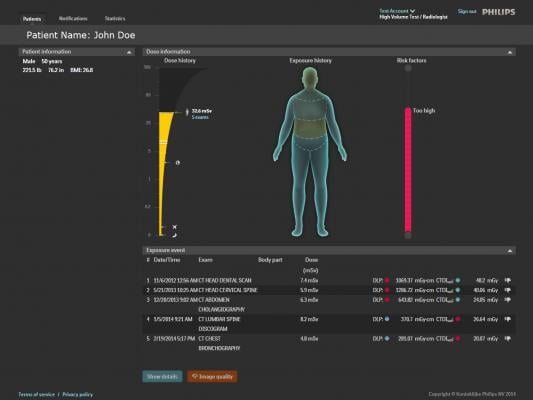
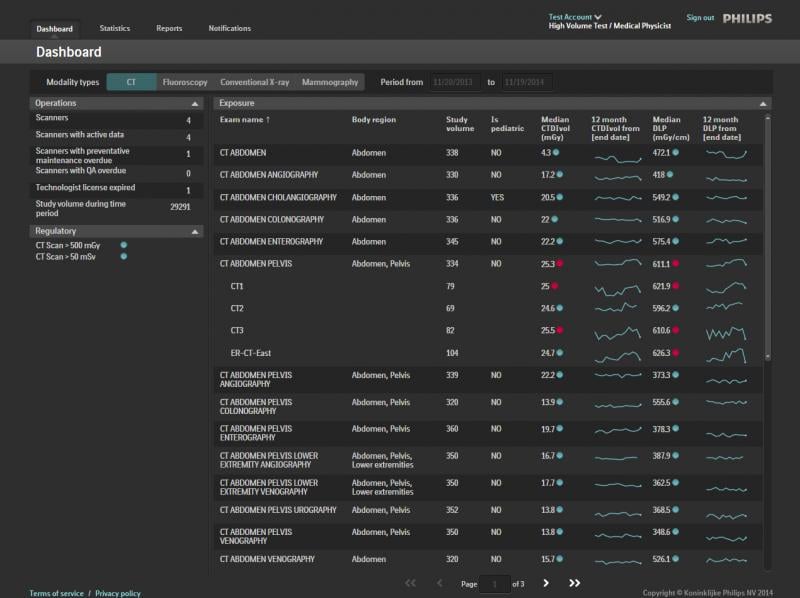
December 12, 2014 — Philips Healthcare introduced the DoseWise Portal, a comprehensive radiation dose management software solution aimed at managing radiation exposure risk to patients and their caregivers at RSNA 2014. Philips DoseWise Portal enables health care providers to proactively record, analyze and monitor imaging radiation dose for patients and clinicians across multiple diagnostic settings.
While the risk of radiation exposure through imaging scans is thought to be low, and the diagnostic rewards generally outweigh those risks, the industry has committed to reducing radiation exposure through improvements in technology and user training. CT scans are of most concern, with a higher average diagnostic radiation dose per scan and nearly 68 million performed annually in the United States
"Dose management is a critical issue, and the reality is that sometimes the higher radiation dose of a CT is necessary for a particular patient to in order reach a definitive diagnosis, in the shortest time, and at the lowest cost," said Gene Saragnese, executive vice president and CEO of Philips Imaging Systems.
Over the past several years, industry standards like MITA XR-29 have provided health care providers with more standardized tools for patient radiation dose management. At the same time, hospitals and health systems looking to better protect patients and clinicians have been forced to tackle the problem independently, using multiple vendors to bring dose management into focus. Philips DoseWise Solutions include a comprehensive portfolio of products and services including ClarityIQ, IMR and DoseAware, that empower health care providers with both the data and support they need to implement a broad and comprehensive dose management strategy.
"Understanding and delivering the right dose for each person each time, and tracking and analyzing exposure over time, requires more than just technology, but also involves a new way managing radiation safety," said Christoph Wald, M.D., Ph.D. and executive vice chair, department of radiology at Lahey Hospital and Medical Center in Burlington, Maryland.
For more information: www.philips.com/healthcare



 May 19, 2026
May 19, 2026 









