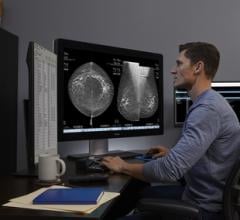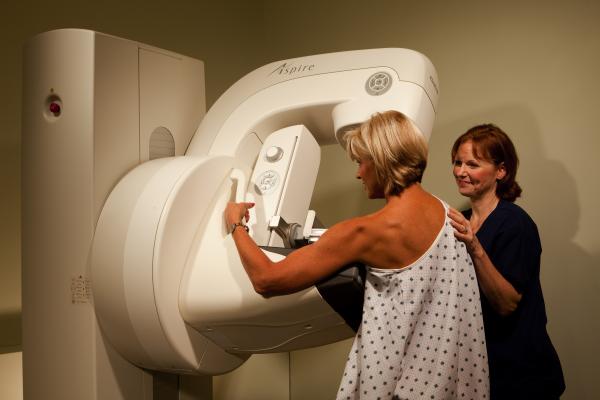
April 9, 2013 — Fujifilm Medical Systems USA, Inc. has introduced its Aspire product line of women’s healthcare solutions.
“The Aspire product line delivers image clarity and patient comfort and is designed to maximize productivity throughout an entire mammography department,” said David Hotchkiss, director of Product Marketing, Women’s Health, Fujifilm Medical Systems USA, Inc. “With Fujifilm, clinicians will acquire exceptional quality images in record time, improving workflow, and making confident diagnostic interpretations.”
The Aspire product family is comprised of a full line of innovative mammography products that meet the needs of any mammography suite including the new Aspire HD Plus, Aspire CRm, Aspire Stereotactic Biopsy, Breast Imaging Diagnostic Workstation and the 1 Shot Phantom M Plus QC toolkit.
For more information: www.fujifilmusa.com/products/medical


 April 20, 2026
April 20, 2026